Sickle Cell Disease and Other Risk Factors for Pediatric Arterial Ischemic Stroke: A Systematic Review and Meta-Analysis
Article information
Abstract
Purpose
The incidence and mortality rates of arterial ischemic stroke (AIS) among pediatric patients have been frequently reported. While pediatric stroke can have multiple severe effects, its risk factors have not been methodically examined. This systematic review and meta-analysis were completed to summarize the existing evidence regarding risk factors for AIS in pediatric patients.
Methods
To gather relevant articles published in the past 15 years, searches were conducted of PubMed and Scopus. Multivariate odds ratios (ORs) and 95% confidence intervals (CIs) were analyzed using Review Manager 5.4.
Results
From the initial screening of 507 articles, five articles comprising a total of 1,423 participants were selected for qualitative analysis. Two of these additionally underwent quantitative analysis. Among the total participants, 1,108 children with AIS (77.9%) had arteriopathy as the underlying disease. Types of arteriopathy included moyamoya disease (24.28%), arterial dissection (23.29%), focal cerebral arteriopathy (16.16%), and vasculitis (14.71%). The meta-analysis revealed that being between 6 and 9 years of age (OR, 2.19; 95% CI, 1.76 to 2.73; P<0.00001) and having sickle cell disease (OR, 3.46; 95% CI, 2.38 to 5.05; P<0.00001) were associated with arteriopathy in pediatric AIS.
Conclusion
Arteriopathy is the most common risk factor for AIS in pediatric patients, and in turn, an age of 6 to 9 years and sickle cell disease are risk factors for arteriopathy within this demographic.
Introduction
A stroke is defined as a sudden onset of focal or global neurological deficit attributed to cerebrovascular injury, such as an infarction or hemorrhage of the central nervous system, that persists for longer than 24 hours [1]. Stroke is the second leading cause of death worldwide [2]. Arterial ischemic stroke (AIS) is responsible for approximately 85% of all strokes, while the remaining cases are due to intracerebral hemorrhage [3].
Stroke can occur at any age. Despite its rarity in children, pediatric stroke is associated with high morbidity and mortality rates, which may stem from delayed diagnosis and a lack of awareness of contributing risk factors [4]. Pediatric stroke is classified into two primary categories based on age. Perinatal stroke is categorized as occurring in neonates, from birth to 28 days of life. In contrast, stroke occurring in children from 29 days to 18 years old is classified as childhood stroke [5-7].
AIS in pediatric patients is a multifactorial disease, influenced by a broad array of risk factors. From 1992 to 2001, the incidence of ischemic stroke among 1,129 pediatric patients was reported to be 1.72 per 100,000 persons annually for childhood stroke and 10.2 per 100,000 live births for perinatal stroke [8]. Separate research published in 2019 reported an incidence of perinatal stroke at 1 per 3,500 live births, along with an incidence of childhood stroke between 1 and 2 per 100,000 persons annually [6]. The in-hospital mortality rate of stroke in children has been reported to account for 2.6% of all pediatric stroke cases [9].
The incidence of AIS in pediatric populations has been widely reported, and this condition can lead to numerous severe consequences. However, its risk factors have not been systematically investigated. Early identification and immediate mitigation of these factors can help minimize morbidity and mortality. This systematic review and meta-analysis were conducted to consolidate the existing evidence on the risk factors for AIS in pediatric populations.
Materials and Methods
The study adhered to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis (PRISMA) guidelines. The design and protocol were registered with PROSPERO (registration number CRD42023454039) on August 24, 2023.
1. Eligibility criteria
Included articles were required to examine the associations of sickle cell disease and other risk factors with AIS in newborns through children up to 18 years old. Additionally, eligible articles had to be observational human studies with retrospective or prospective cohort designs that were published within the prior 15 years and had a minimum sample size of 10. To be selected for meta-analysis, articles needed to provide odds ratio (OR) and 95% confidence interval (CI) values. Case reports, literature reviews, systematic reviews, meta-analyses, animal studies, and articles without full text available were excluded.
2. Data sources and search strategy
Articles were systematically searched in PubMed and Scopus, using a combination of free-text searching and keyword probing. The search terms employed were as follows: ("risk factor" OR "risk factors" OR "predictor" OR "predictors") AND ("stroke" OR "ischemic stroke" OR "arterial ischemic stroke") AND ("pediatric" OR "neonates" OR "perinatal" OR "childhood" OR "children"). The reference lists from the resulting articles were manually reviewed to identify additional relevant studies.
3. Data selection and quality assessment
Four authors (CPT, LW, EHT, and AMG) manually screened the titles and abstracts based on the eligibility criteria. The full texts of the selected articles underwent a second round of evaluation. Additionally, the Newcastle-Ottawa scale (NOS) instrument was employed to assess the articles. In instances involving disagreement, the fifth author (NMDP) was consulted to facilitate a consensus.
4. Data extraction and statistical analysis
The data from the chosen articles were extracted by four authors (CPT, LW, EHT, and AMG) as follows: first author, publication date, country, population, sample size, and outcomes. The statistical analysis for the meta-analysis was conducted using Review Manager (RevMan) version 5.4 (Stata Corp., College Station, TX, USA).
Results
1. Study selection
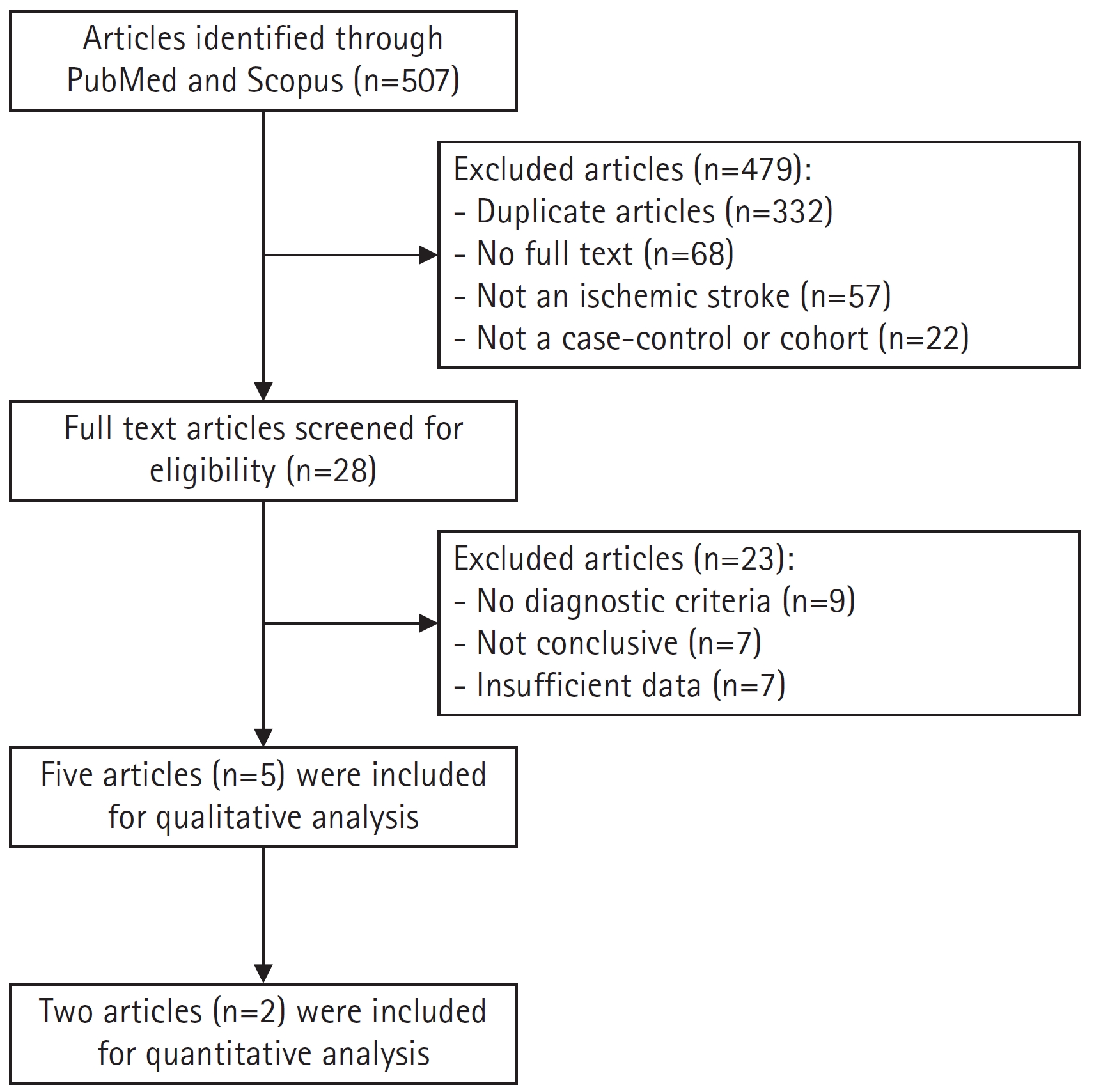
A total of 507 articles were identified through searches of PubMed and Scopus. The initial screening led to the exclusion of 479 articles, which included 332 duplicates, 68 articles without full text, 57 articles in which the variable was not ischemic stroke, and 22 articles that were neither case-control nor cohort studies. The second screening involved a full-text review of the remaining 28 articles to determine their eligibility, resulting in the exclusion of 23 articles. Ultimately, five articles were deemed suitable for the systematic review, and two were included in the meta-analysis (Fig. 1).
2. Quality assessment
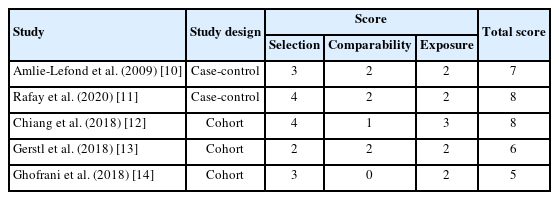
The quality of each article was evaluated using the NOS instrument, as shown in Table 1 [10-14]. Articles were categorized based on their total scores: a score of 7 to 10 indicated high quality, a score of 4 to 6 suggested a low risk of bias, and a score of 1 to 3 denoted a high risk of bias. Among the articles assessed, two received a total score of 8 out of 10, one scored 7, one scored 6, and one scored 5. Thus, three articles were deemed high quality, while two were considered to have a low risk of bias.
3. Study characteristics and qualitative analysis
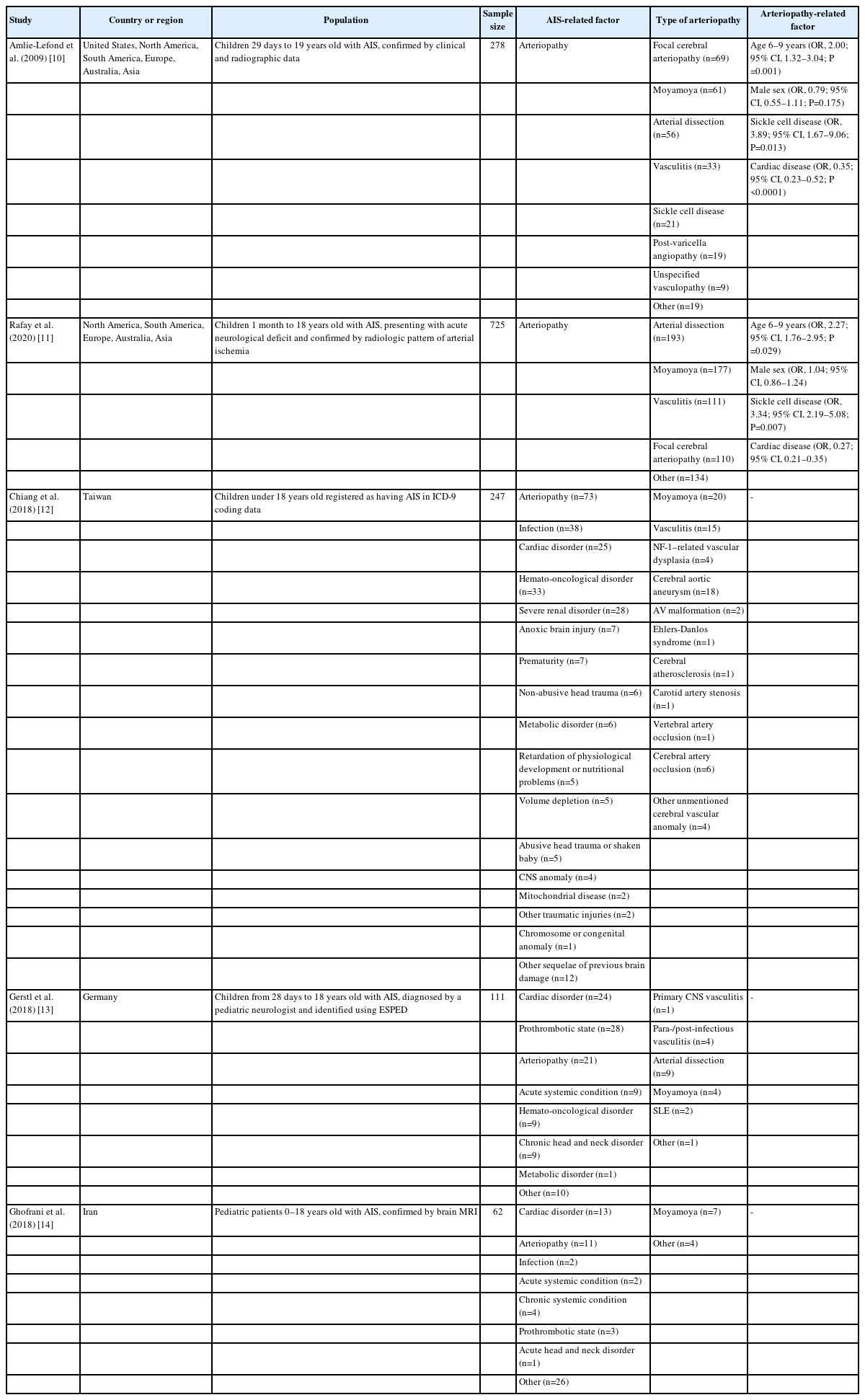
The articles included in this review were published between 2009 and 2020 and encompass two case-control studies and three cohort studies, as detailed in Table 2. These studies were conducted across various regions, including North America, South America, Europe, Australia, Asia, the United States, Taiwan, Germany, and Iran, and involved a total of 1,423 participants. The participant population in three of the studies consisted of children ranging in age from 28–30 days to 18–19 years, while the remaining two studies focused on neonates and children under the age of 18. The diagnostic criteria for AIS were derived as follows: two articles utilized clinical neurological deficit and radiological data from the International Pediatric Stroke Study; one article employed the International Classification of Diseases, Ninth Revision coding system; another article relied on diagnoses from pediatric neurologists and the German Paediatric Surveillance Unit; and one article based its assessments on brain magnetic resonance imaging.
In the review of five studies involving 1,423 pediatric patients with AIS, the underlying diseases were found to be arteriopathy (77.9%), cardiac disorder (4.36%), hemato-oncological disorder (2.95%), infection (2.81%), and prothrombotic state (2.18%). Other conditions were also identified, including severe renal disorder (1.97%), sequelae of previous brain damage (0.84%), acute systemic condition (0.77%), chronic head and neck disorder (0.63%), anoxic brain injury (0.49%), prematurity (0.49%), metabolic disorder (0.49%), non-abusive head trauma (0.42%), retardation of physiological development or nutritional problems (0.35%), volume depletion (0.35%), abusive head trauma or shaken baby syndrome (0.35%), central nervous system anomaly (0.28%), chronic systemic condition (0.28%), mitochondrial disease (0.14%), other traumatic injuries (0.14%), chromosomal or congenital anomaly (0.07%), acute head and neck disorder (0.07%), and other unspecified conditions (2.53%).
In a study of 1,108 pediatric patients with AIS, the types of arteriopathy identified were moyamoya (24.28%), arterial dissection (23.29%), focal cerebral arteriopathy (16.16%), and vasculitis (14.71%). Other arteriopathies detected included sickle cell disease (1.9%), post-varicella angiopathy (1.71%), cerebral aortic aneurysm (1.62%), cerebral artery occlusion (0.54%), neurofibromatosis type I-related vascular dysplasia (0.36%), systemic lupus erythematosus (0.18%), arteriovenous malformation (0.18%), other peripheral vascular anomalies (0.18%), Ehlers-Danlos syndrome (0.09%), cerebral atherosclerosis (0.09%), carotid artery stenosis (0.09%), vertebral artery occlusion (0.09%), and other types of arteriopathy not specified (14.62%).
4. Quantitative analysis
Two articles with the same outcome variables were combined for meta-analysis. This meta-analysis examined the associations between an age of 6 to 9 years, male sex, sickle cell disease, and cardiac disease in relation to arteriopathy in pediatric AIS. As depicted in Fig. 2, an age between 6 and 9 years (OR, 2.19; 95% CI, 1.76 to 2.73; P<0.00001) and sickle cell disease (OR, 3.46; 95% CI, 2.38 to 5.05; P<0.00001) were significantly associated with AIS in pediatric patients. Arteriopathy in pediatric AIS was found to have a significant negative association with cardiac disease (OR, 0.29; 95% CI, 0.23 to 0.35; P<0.00001); however, no significant association was found with male sex (OR, 0.98; 95% CI, 0.83 to 1.15; P=0.77).
Discussion
Pediatric AIS is a multifactorial disease with a variety of underlying causes, some of which may have been previously unrecognized as contributing factors. Rather than focusing solely on therapeutic interventions for the disease, identifying these risk factors can serve as an effective strategy to decrease its incidence. Early recognition of risk factors presents opportunities to reduce both the mortality and morbidity rates associated with pediatric AIS.
The qualitative analysis presented in Table 2 reveals a variety of underlying diseases associated with pediatric AIS. Of these, arteriopathy emerged as the most frequently occurring disease across the five included studies. More specifically, moyamoya disease was identified as the most common type of arteriopathy in pediatric AIS. Moyamoya disease is a chronic cerebrovascular condition of unknown origin, characterized by enduring stenosis of the intracranial blood vessels. It most commonly affects the terminal portion of the internal carotid and middle cerebral arteries [15]. This occlusion of the large intracranial artery contributes to hemodynamic disturbances, resulting in hypoperfusion that can induce hypoxic injuries in brain cells [16].
Artery dissection is the second most common type of arteriopathy. This condition arises when a tear forms within the inner lining of the arterial wall. The compromised structure of the inner arterial vessel allows blood flow to reach these torn areas, which may lead to clot formation. These unstable clots can disrupt blood circulation, including that of the brain, thereby interfering with cerebral perfusion and potentially resulting in ischemic stroke [7,17]. Notably, several medical conditions (such as Ehlers-Danlos syndrome, vasculitis, and head or neck trauma) that have been identified as underlying diseases and other types of arteriopathy in pediatric AIS have also been associated with arterial dissection [6,9,18-20].
The quantitative analysis presented in Fig. 2 indicates an association between sickle cell disease and AIS in pediatric patients. Sickle cell disease is a hereditary disorder resulting from a defect in the hemoglobin gene on chromosome 11, specifically the beta-globin component [21]. This single base-pair point mutation disrupts the function of hemoglobin, a molecule within erythrocytes responsible for delivering oxygen to cells [22]. In sickle cell anemia, the most common form of sickle cell disease, severe anemia can lead to a reduction in oxygen perfusion to the brain [23]. Vasculopathy is also predominantly observed in patients with sickle cell disease [24]. Furthermore, sickle cell disease is associated with an elevated risk of thrombophilia, which can disrupt the delivery of oxygen to brain tissue [25].
Children between the ages of 6 and 9 years have been found to have a heightened risk of AIS compared to other age groups [10,11]. This finding may be attributed to the fact that children aged 6 years and older tend to develop more advanced motor skills, which carry a higher risk of trauma compared to the motor skills of younger children. These skills include activities such as riding a two-wheeled bicycle and other tasks that require coordination with larger muscles. Furthermore, children aged 9 years and under typically have not yet developed stable self-control [26,27]. The ability to control and regulate impulsivity is age-dependent and aligns with the maturation of the prefrontal cortex, the center for impulse control. This maturation generally occurs in children aged 10 years and above [28]. Consequently, these factors contribute to children aged 6 to 9 years exhibiting an elevated risk of traumatic brain injury, which is a risk factor for AIS in pediatric populations [12,29].
A negative correlation was identified between a history of cardiac disease and arteriopathy in pediatric AIS, based on the combined OR results of two included studies. Children with cardiac disease were less likely to experience AIS than children without cardiac disease. This is likely due to incomplete vascular imaging in both cardiac and non-cardiac patients, as detailed in the included studies [10,11]. However, these results should be interpreted with caution due to the potential use of antiplatelet agents in patients with cardiac disease. Research indicates that antiplatelet agents can reduce the risk of AIS in pediatric patients [30]. In contrast, cardiac disorder has been found to increase the risk of pediatric AIS [12-14,31-34]. Future research on the relationship between cardiac disease and AIS in pediatric patients should be more specific, focusing on diagnostic investigation, medication history, and other potentially influential factors.
In alignment with previous studies, the findings did not reveal a significant association between male sex and pediatric AIS [12,33,35]. However, other research has identified a significant male predominance in the incidence of AIS in pediatric populations [7,13,36,37]. Further studies are needed to explore the relationship between sex and pediatric AIS, including potential underlying mechanisms.
This study provides evidence of risk factors for AIS in pediatric populations, based on both qualitative analysis and quantitative analysis with a high statistical conclusion value. However, this study had several limitations due to confounding factors. Notably, the analysis included a small number of studies, which resulted from the limited number of updated articles discussing pediatric AIS over the past decade. Future research on pediatric AIS should be conducted with a larger population. Additionally, further studies should be performed to explore the association between existing risk factors and AIS in pediatric patients to uncover the underlying mechanism.
In conclusion, arteriopathy is the most common risk factor for AIS in pediatric patients. The age range of 6 to 9 years, along with the presence of sickle cell disease, are the primary risk factors for arteriopathy in pediatric AIS.
Notes
No potential conflict of interest relevant to this article was reported.
Author contribution
Conceptualization: CPT and LW. Data curation: CPT, LW, EHT, and AMG. Formal analysis: CPT, LW, EHT, AMG, and NMDP. Methodology: CPT and LW. Writing-original draft: CPT. Writing-review & editing: CPT, LW, EHT, AMG, and NMDP.
Acknowledgements
The authors express their gratitude to the Faculty of Medicine at Widya Mandala Catholic University Surabaya in Surabaya, Indonesia, as well as the Department of Neurology at the Faculty of Medicine, Udayana University in Bali, Indonesia, for their support.