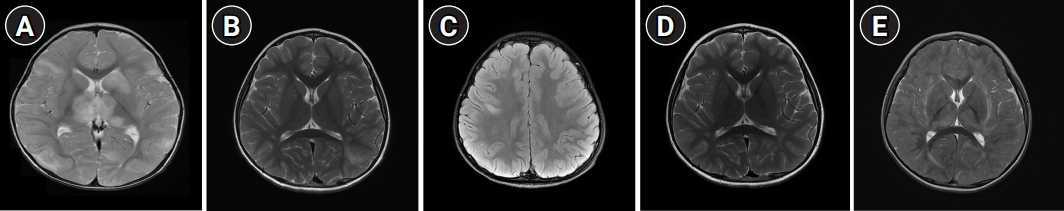
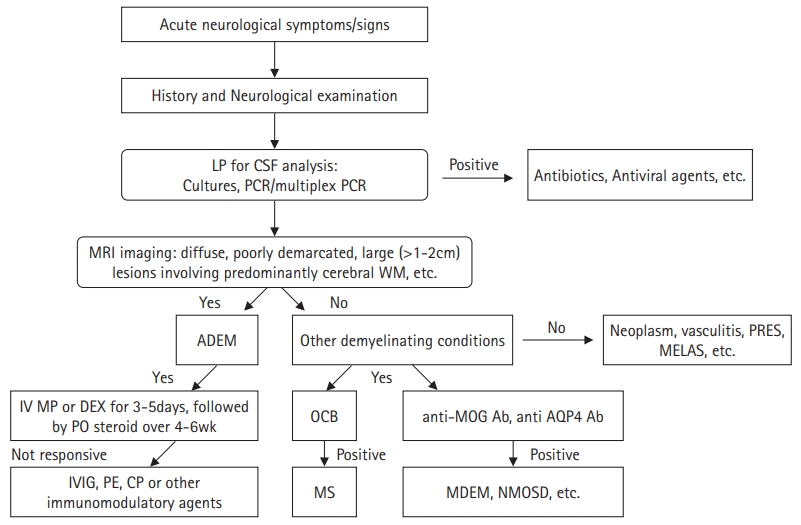
1. Cole J, Evans E, Mwangi M, Mar S. Acute disseminated encephalomyelitis in children: an updated review based on current diagnostic criteria. Pediatr Neurol 2019;100:26-34.


6. Pohl D, Alper G, Van Haren K, Kornberg AJ, Lucchinetti CF, Tenembaum S, et al. Acute disseminated encephalomyelitis: updates on an inflammatory CNS syndrome. Neurology 2016;87(9 Suppl 2):S38-45.


8. Hacohen Y, Mankad K, Chong WK, Barkhof F, Vincent A, Lim M, et al. Diagnostic algorithm for relapsing acquired demyelinating syndromes in children. Neurology 2017;89:269-78.


9. Hahn CD, Miles BS, MacGregor DL, Blaser SI, Banwell BL, Hetherington CR. Neurocognitive outcome after acute disseminated encephalomyelitis. Pediatr Neurol 2003;29:117-23.


10. Anlar B, Basaran C, Kose G, Guven A, Haspolat S, Yakut A, et al. Acute disseminated encephalomyelitis in children: outcome and prognosis. Neuropediatrics 2003;34:194-9.


11. Christensen PS, Ostergaard JR. Acute disseminated encephalomyelitis: definition, treatment, prognosis and evidence. Ugeskr Laeger 2008;170:1839-42.

15. Wan Sulaiman WA, Inche Mat LN, Hashim HZ, Hoo FK, Ching SM, Vasudevan R, et al. Acute disseminated encephalomyelitis in dengue viral infection. J Clin Neurosci 2017;43:25-31.


16. Abe T, Kojima K, Shoji H, Tanaka N, Fujimoto K, Uchida M, et al. Japanese encephalitis. J Magn Reson Imaging 1998;8:755-61.


17. Bhardwaj U, Pandey N, Rastogi M, Singh SK. Gist of Zika virus pathogenesis. Virology 2021;560:86-95.


18. Garg RK. Spectrum of neurological manifestations in COVID-19: a review. Neurol India 2020;68:560-72.


21. Etemadifar M, Mansouri AR, Nouri H, Sedaghat N, Salari M, Maghsoudi M, et al. Post-COVID-19 acute disseminated encephalomyelitis: case report and review of the literature. Neuroimmunol Rep 2022;2:100066.

22. Karussis D, Petrou P. The spectrum of post-vaccination inflammatory CNS demyelinating syndromes. Autoimmun Rev 2014;13:215-24.


23. Alves-Leon SV, Veluttini-Pimentel ML, Gouveia ME, Malfetano FR, Gaspareto EL, Alvarenga MP, et al. Acute disseminated encephalomyelitis: clinical features, HLA DRB1*1501, HLA DRB1*1503, HLA DQA1*0102, HLA DQB1*0602, and HLA DPA1*0301 allelic association study. Arq Neuropsiquiatr 2009;67(3A):643-51.


26. Tenembaum S, Chitnis T, Ness J, Hahn JS; International Pediatric MS Study Group. Acute disseminated encephalomyelitis. Neurology 2007;68(16 Suppl 2):S23-36.


28. Ishizu T, Minohara M, Ichiyama T, Kira R, Tanaka M, Osoegawa M, et al. CSF cytokine and chemokine profiles in acute disseminated encephalomyelitis. J Neuroimmunol 2006;175:52-8.


29. Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol 2005;23:683-747.


30. Probstel AK, Dornmair K, Bittner R, Sperl P, Jenne D, Magalhaes S, et al. Antibodies to MOG are transient in childhood acute disseminated encephalomyelitis. Neurology 2011;77:580-8.


31. Tenembaum SN. Pediatric demyelinating disease and antiŌĆÉMOG antibody. Clin Exp Neuroimmunol 2021;12:7-21.


33. Hennes EM, Baumann M, Schanda K, Anlar B, Bajer-Kornek B, Blaschek A, et al. Prognostic relevance of MOG antibodies in children with an acquired demyelinating syndrome. Neurology 2017;89:900-8.


35. Baum PA, Barkovich AJ, Koch TK, Berg BO. Deep gray matter involvement in children with acute disseminated encephalomyelitis. AJNR Am J Neuroradiol 1994;15:1275-83.


36. Wong YY, van Pelt ED, Ketelslegers IA, Catsman-Berrevoets CE, Hintzen RQ, Neuteboom RF, et al. Evolution of MRI abnormalities in paediatric acute disseminated encephalomyelitis. Eur J Paediatr Neurol 2017;21:300-4.


37. Honkaniemi J, Dastidar P, Kahara V, Haapasalo H. Delayed MR imaging changes in acute disseminated encephalomyelitis. AJNR Am J Neuroradiol 2001;22:1117-24.


38. Takahashi T, Fujihara K, Nakashima I, Misu T, Miyazawa I, Nakamura M, et al. Anti-aquaporin-4 antibody is involved in the pathogenesis of NMO: a study on antibody titre. Brain 2007;130(Pt 5):1235-43.


39. Arktout S. Prognosis factors in children with ADEM: clinical, biological, and radiological features. Int J Radiol Imaging Technol 2020;6:063.

41. Pavone P, Pettoello-Mantovano M, Le Pira A, Giardino I, Pulvirenti A, Giugno R, et al. Acute disseminated encephalomyelitis: a long-term prospective study and meta-analysis. Neuropediatrics 2010;41:246-55.


43. Dale RC, de Sousa C, Chong WK, Cox TC, Harding B, Neville BG. Acute disseminated encephalomyelitis, multiphasic disseminated encephalomyelitis and multiple sclerosis in children. Brain 2000;123 Pt 12:2407-22.


44. Hynson JL, Kornberg AJ, Coleman LT, Shield L, Harvey AS, Kean MJ. Clinical and neuroradiologic features of acute disseminated encephalomyelitis in children. Neurology 2001;56:1308-12.


45. Sahlas DJ, Miller SP, Guerin M, Veilleux M, Francis G. Treatment of acute disseminated encephalomyelitis with intravenous immunoglobulin. Neurology 2000;54:1370-2.


46. RamachandranNair R, Rafeequ M, Girija AS. Plasmapheresis in childhood acute disseminated encephalomyelitis. Indian Pediatr 2005;42:479-82.

47. Miyazawa R, Hikima A, Takano Y, Arakawa H, Tomomasa T, Morikawa A. Plasmapheresis in fulminant acute disseminated encephalomyelitis. Brain Dev 2001;23:424-6.