 |
 |
- Search
| Ann Child Neurol > Volume 31(4); 2023 > Article |
|
Abstract
Purpose
Methods
Results
Conclusion
Supplementary materials
Supplementary┬ĀFig.┬Ā1.
Notes
Author contribution
Conceptualization: JYH and JMK. Data curation: HY and JMK. Formal analysis: JYH and JMK. Funding acquisition: JMK. Methodology: JYH. Project administration: JMK. Visualization: JYH. Writing-original draft: HY and JMK. Writing-review & editing: HY, JYH, and JMK.
Acknowledgments
Fig.┬Ā1.
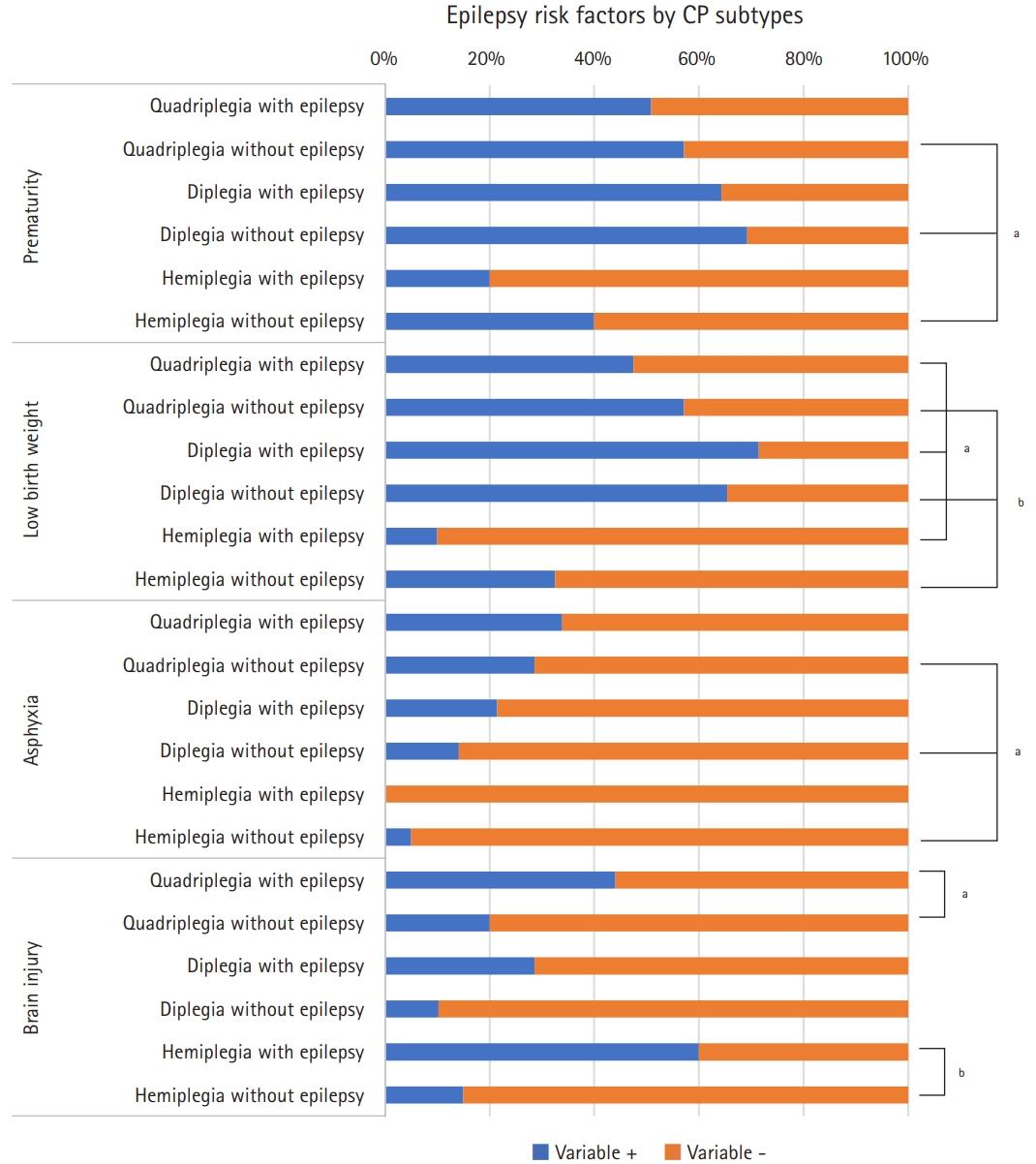
Fig.┬Ā2.
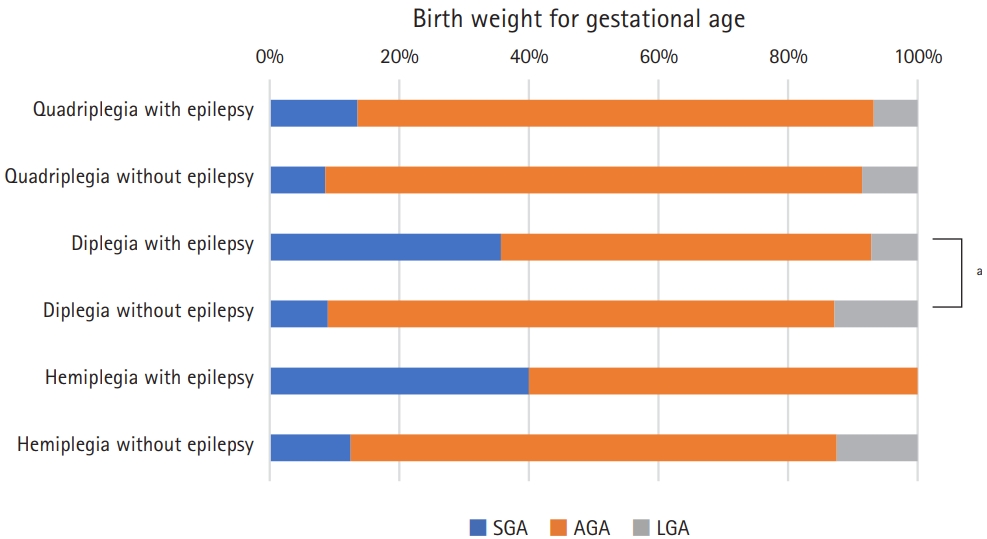
Fig.┬Ā3.
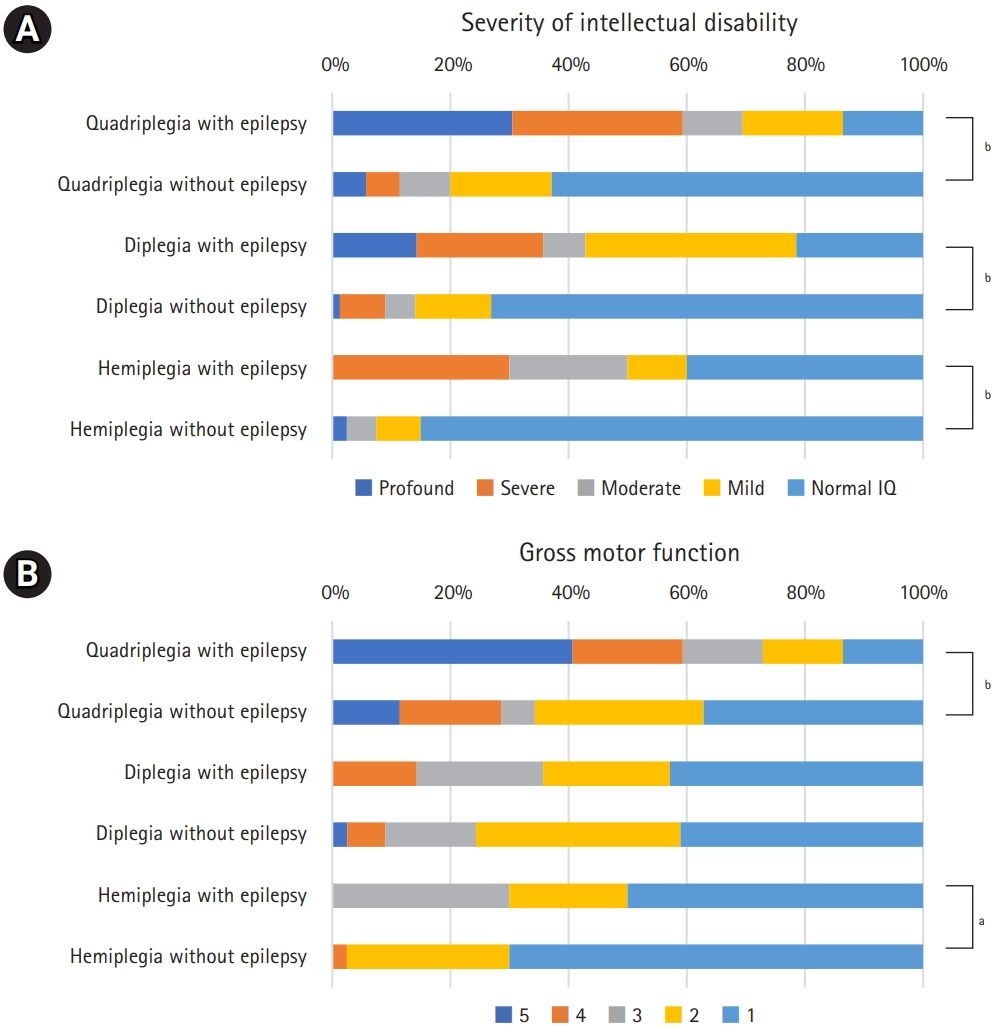
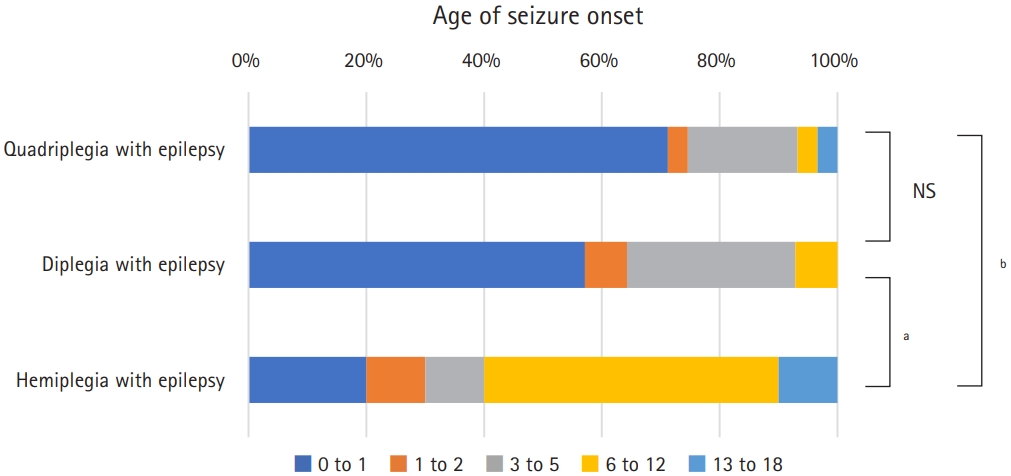
Fig.┬Ā4.
Table┬Ā1.
Table┬Ā2.
Values are presented as number (%).
CP, cerebral palsy; IUGR, intrauterine growth retardation; NICU, neonatal intensive care unit; GMFCS-E&R, Gross Motor Function Classification System-Expanded and Revised; IQ, intellectual quotient; PVL, periventricular leukomalacia; PVWMI, periventricular white matter injury; GMI, gray matter injury.
aPercentage among patients with available data.
Table┬Ā3.
Values are presented as number (%).
IUGR, intrauterine growth retardation; NICU, neonatal intensive care unit; GMFCS-E&R, Gross Motor Function Classification System-Expanded and Revised; IQ, intellectual quotient; ED, epileptiform discharge; PVL, periventricular leukomalacia; PVWMI, periventricular white matter injury; GMI, gray matter injury.
aPercentage among patients with available data.
Table┬Ā4.
References
- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 1,924 View
- 82 Download
- Related articles in Ann Child Neurol
-
Status Epilepticus in Children: Experience in a Portuguese Tertiary Hospital2023 July;31(3)
Clinical and Genetic Characteristics of Young Children with Fragile X Syndrome2021 July;29(3)






