|
 |
- Search
| Ann Child Neurol > Volume 31(3); 2023 > Article |
|
Tramadol is a recently developed, centrally acting synthetic analgesic agent. The pharmacological mechanism of tramadol has not yet been fully elucidated, but unlike standard opioid analgesics, tramadol alleviates pain by modulating norepinephrine secretion and inhibiting serotonin reuptake [1]. The potency of tramadol is between 10% and 25% of that of morphine at the ╬╝-opioid receptor, so it is considered a ŌĆ£weak opioid.ŌĆØ For this reason, it is regarded as relatively safe. Tramadol has been commonly used for postoperative pain treatment in children who have mild to moderate pain. The recommended dose for the intravenous (IV) route for children is 2 mg/kg every 4 to 6 hours, which is best for analgesic action with minimal side effects [2].
However, tramadol has some unique properties compared to other standard opioid medications that are attributable to its mechanism of inhibiting monoamine reuptake. Examples of the adverse effects of tramadol include respiratory depression, seizure, tachycardia, hypertension, serotonin syndrome, and manic syndrome [3].
Seizures have been noted as a concerning side effect of tramadol since its market approval in the United States in 1995, based on post-marketing reports to the U.S. Food and Drug Administration (FDA). Between 1997 and 2017, 30,730 tramadol-related cases had been reported to the FDA's Adverse Event Reporting System, and seizures accounted for 7% of the cases [4].
The FDA issued a black-box warning in 2017, banning the use of tramadol in children and adolescents under the age of 12, and in those aged 12 to 18 with underlying diseases, due to its potential to cause serious respiratory side effects and death [5]. Despite the FDAŌĆÖs warning, tramadol continues to be given, which is worrisome.
Here, we present a case of tramadol-induced status epilepticus in a 15-year-old girl with no past history of seizures. A 15-year-old girl (height, 159 cm; weight, 47 kg) presented to the emergency department due to abdominal pain. She had normal developmental milestones and her past medical history was unremarkable.
She had undergone laparoscopic left ovarian cystectomy (pathology: functional cyst) 23 days earlier. A clinical examination revealed normal hemodynamic variables, and there was no sign of dehydration, or fever. On physical examination, widespread abdominal tenderness without rebound tenderness was found, while other physical examination and lab test results were normal. Abdominopelvic computed tomography (CT) showed paralytic small bowel ileus. She was referred to the gynecology department for conservative management. The patient was given 30 mg of IV ketorolac tromethamine (Trolac, Whanin Pharm Co., Seoul, Korea) and 100 mg of IV tramadol (Tandol, AJU Pharm Co., Seoul, Korea). Since the pain persisted, IV tramadol was administered at intervals of 2 hours, shorter than the recommended interval, totaling 200 mg. About 1 hour after the second dose of IV tramadol administration, she developed a symmetric and generalized tonic seizure. The seizure lasted for 20 minutes and was suppressed with 4 mg of IV lorazepam. After approximately 13 hours, a focal tonic seizure of the right hand and leg with impaired awareness developed. The seizure was immediately suppressed with an additional administration of IV lorazepam, and she was referred to the pediatric neurology department for further management.
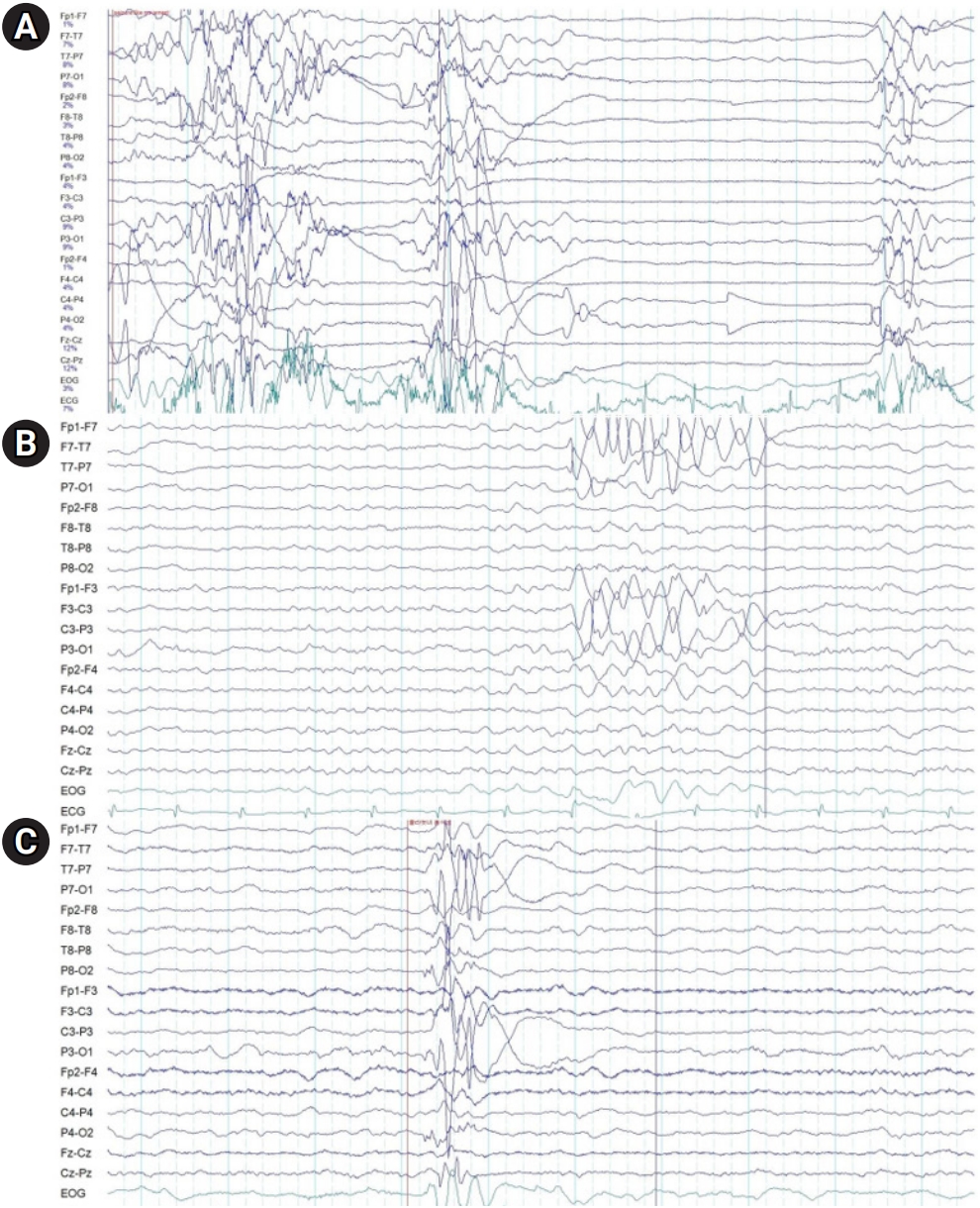
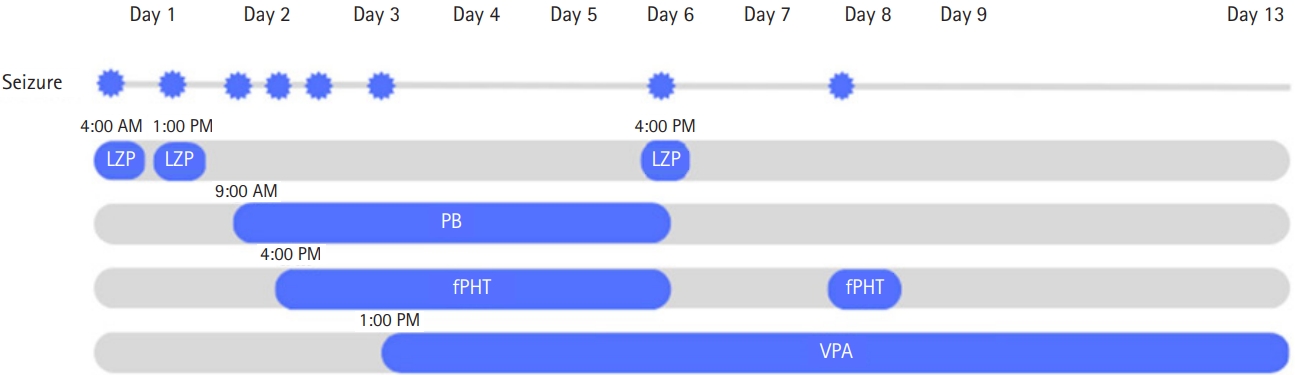
On the 2nd day of hospitalization, at 8:00 AM, a tonic seizure of both arms was observed and IV phenobarbital loading (20 mg/kg) was started. In order to rule out intracranial hemorrhage or any intracranial lesion, brain CT and brain magnetic resonance imaging (MRI) were performed and the results showed no abnormalities. An electroencephalogram (EEG) was performed and demonstrated slow background activity with theta bursts of 6 to 7 Hz sharp waves over the left hemisphere (Fig. 1A). At 4:00 PM on the same day, another tonic seizure of both arms was observed and IV fosphenytoin loading (20 mg phenytoin [PE]/kg) was started. At midnight, the patient woke up disoriented with crying, and she calmed down after 30 minutes. On the 3rd day of hospitalization, she first reported light headache and dizziness, followed by a generalized tonic-clonic seizure with vocalization. IV valproate was added. On the 5th day of hospitalization, a follow-up EEG was obtained and did not document significant changes. At 4:00 PM on the 6th day of hospitalization, the patient had a paroxysmal attack and cried out sharply while being emotionally distressed, agitated, and cognitively disoriented. Additionally, aggressive behaviors and impaired speech were observed. The event lasted for 20 minutes and was suppressed with 4 mg of IV lorazepam. IV phenobarbital and IV fosphenytoin were discontinued because of their unconfirmed effectiveness. At midnight on the 8th day of hospitalization, she developed a symmetric and generalized tonic seizure that lasted for 20 minutes, and IV fosphenytoin half-loading (10 mg PE/kg) was added again. The patient was given multiple antiseizure medications: lorazepam, phenobarbital, fosphenytoin, and valproic acid (Fig. 2). Follow-up EEG showed progressive reduction of theta bursts and suppressed background activity (Fig. 1B and C). On the 9th day of hospitalization, she showed no seizures and IV fosphenytoin was stopped. The patient was fully oriented and awake, and no further seizures or paroxysmal activities were noted. On the 13th day of hospitalization, she was discharged without any neurologic sequelae, and oral valproic acid was continued for an additional 4-week period. In the outpatient clinic, the patient exhibited normal EEG results after the discontinuation of valproic acid. No seizure was reported during 6 months of follow-up.
The patientŌĆÖs legal caregiver provided written informed consent for the publication of the patientŌĆÖs clinical details and other related information. This study was approved by the Institutional Review Board (IRB) of Daejin Medical Center (IRB No. 2023-01-006).
Seizures are a side effect of tramadol. This epileptogenic effect of tramadol occurs at both low and high doses [6]. The Adverse Drug Reactions Advisory Committee reported that tramadol can cause seizures, and using other drugs that lower the convulsive threshold concurrently could increase the likelihood of convulsions [7]. It is thought that the combination of injections at shorter-than-recommended intervals and the concurrent use of ketorolac may have exacerbated the side effects.
Talaie et al. [6] studied 61 patients with tramadol-induced seizures and reported normal results of EEG and CT scans of the brain in most patients. Our patient had normal results of brain CT and MRI, but abnormal EEG results. These abnormalities gradually decreased over time, and an EEG performed in the outpatient clinic returned to normal.
More problematically, the interpretation of repeated seizures despite discontinuing tramadol use and poor response to anti-seizure medication (ASM) remains. The patient was treated with multiple ASMs, but there was no prompt resolution of the seizures, and convulsive events repeated for 8 days. Further research on this pattern is deemed necessary.
In 2017, the FDA issued a black-box warning against the use of tramadol in children; however, there remains a population of children who continue to receive tramadol. In line with previous studies, the current case study suggests that a therapeutic dose of tramadol can induce seizures and even cause status epilepticus. Careful consideration is required when administering tramadol to adolescents.
Notes
Author contribution
Conceptualization: SYK and SHB. Data curation: SYK and YEK. Methodology: SYK and YEK. Project administration: SHB. Visualization: SYK and KWK. Writing - original draft: SYK. Writing - review & editing: SHB.
References
1. Schug SA. The role of tramadol in current treatment strategies for musculoskeletal pain. Ther Clin Risk Manag 2007;3:717-23.


3. Beakley BD, Kaye AM, Kaye AD. Tramadol, pharmacology, side effects, and serotonin syndrome: a review. Pain Physician 2015;18:395-400.


4. U.S. Food and Drug Administration. FDA Adverse Event Reporting System [Internet]. Silver Spring: FDA; 2023 [cited 2023 May 16]. Available from: https://open.fda.gov/data/faers
5. U.S. Food and Drug Administration. FDA Drug Safety Communication. FDA restricts use of prescription codeine pain and cough medicines and tramadol pain medicines in children: recommends against use in breastfeeding women [Internet]. Silver Spring: FDA; 2018 [cited 2023 May 16]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-restricts-use-prescription-codeine-pain-and-cough-medicines-and
6. Talaie H, Panahandeh R, Fayaznouri M, Asadi Z, Abdollahi M. Dose-independent occurrence of seizure with tramadol. J Med Toxicol 2009;5:63-7.




7. Adverse Drug Reactions Advisory Committee. Tramadol: four yearsŌĆÖ experience. Aust Adv Drug React Bull 2003;22:2-3.
- TOOLS
-
METRICS

-
- 1 Crossref
- Scopus
- 1,727 View
- 75 Download
- Related articles in Ann Child Neurol
-
A Case of Encephalitis Caused by Epstein-Barr Virus Infection.2009 November;17(2)
A Case of a Child with Seizure caused by Organophosphate Poisoning.2018 September;26(3)
The Clinical Characteristics of Status Epilepticus in Children.1999 October;7(1)