|
 |
- Search
| Ann Child Neurol > Volume 31(1); 2023 > Article |
|
Neonatal hypoglycemia is a common condition that constitutes one of the leading causes of brain injury. Symptomatic hypoglycemia may present with metabolic encephalopathy characterized by neurological features, including stupor, jitteriness, seizures, apnea, irritability, and muscular hypotonia mimicking several other diseases. The typical magnetic resonance imaging (MRI) patterns of neonatal hypoglycemic encephalopathy (NHE) are signal abnormalities localized to the parietal and occipital lobes and progressive parenchymal loss of the predominant occipital lobe in the newborn. Diffusion-weighted imaging (DWI) may show restricted diffusion in the involved areas. These brain lesions are transient, and the reversibility of DWI has also been reported [1]. The patterns of injury associated with NHE are more diverse than those previously reported, and include white matter, cortical, and basal ganglia/thalamic abnormalities, white matter hemorrhage, and middle cerebral artery territory infarctions [2]. However, no reports of NHE with extensive white matter involvement and serial changes on MRI are available. Here, we present a case of NHE with atypical MRI findings along with sequential neuroradiological changes. This study was approved by the Institutional Review Board of Daegu Catholic University Medical Center (CR-22-014). The requirement for publication consent was waived because personally identifiable protected health information was not disclosed in this report.
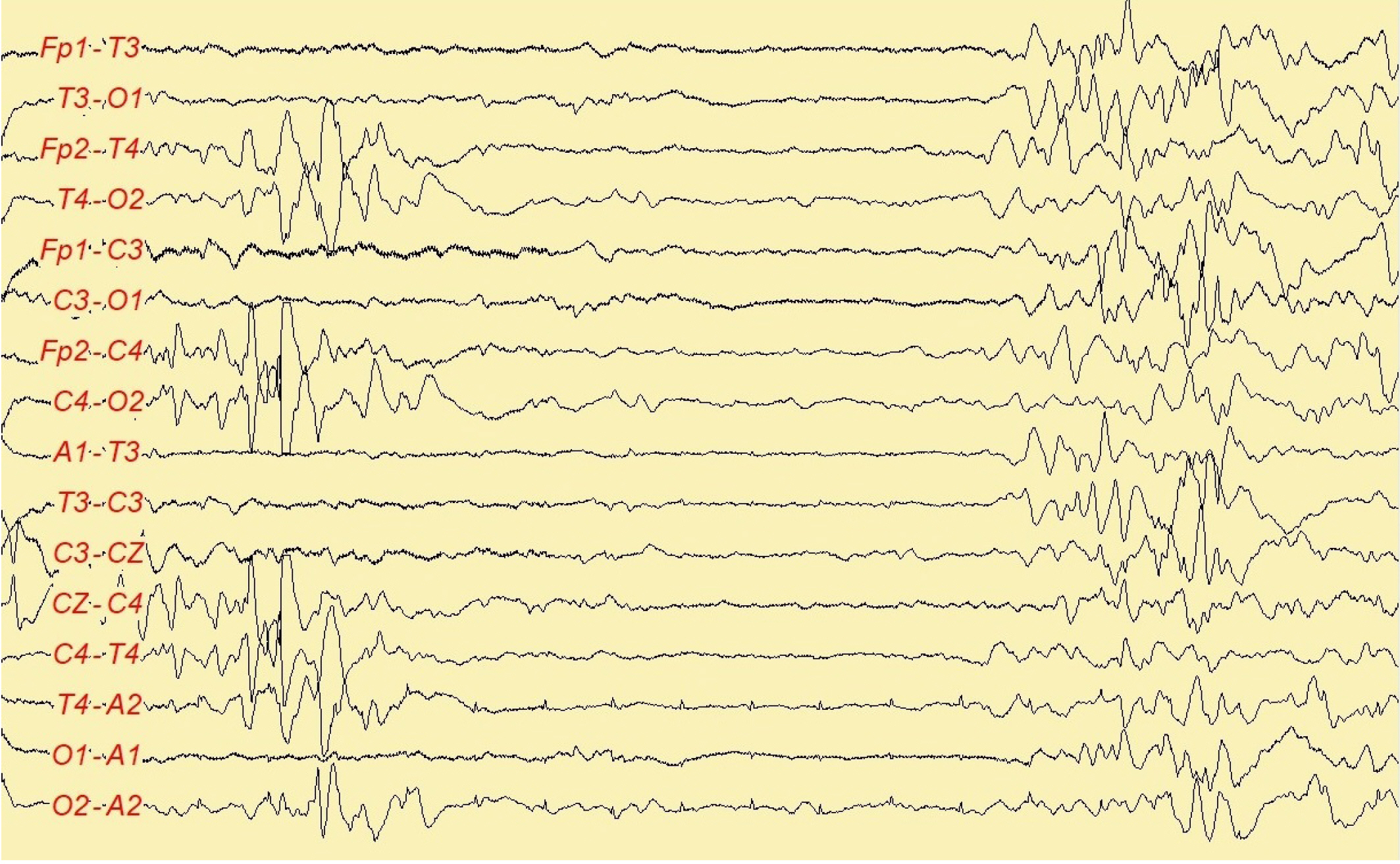
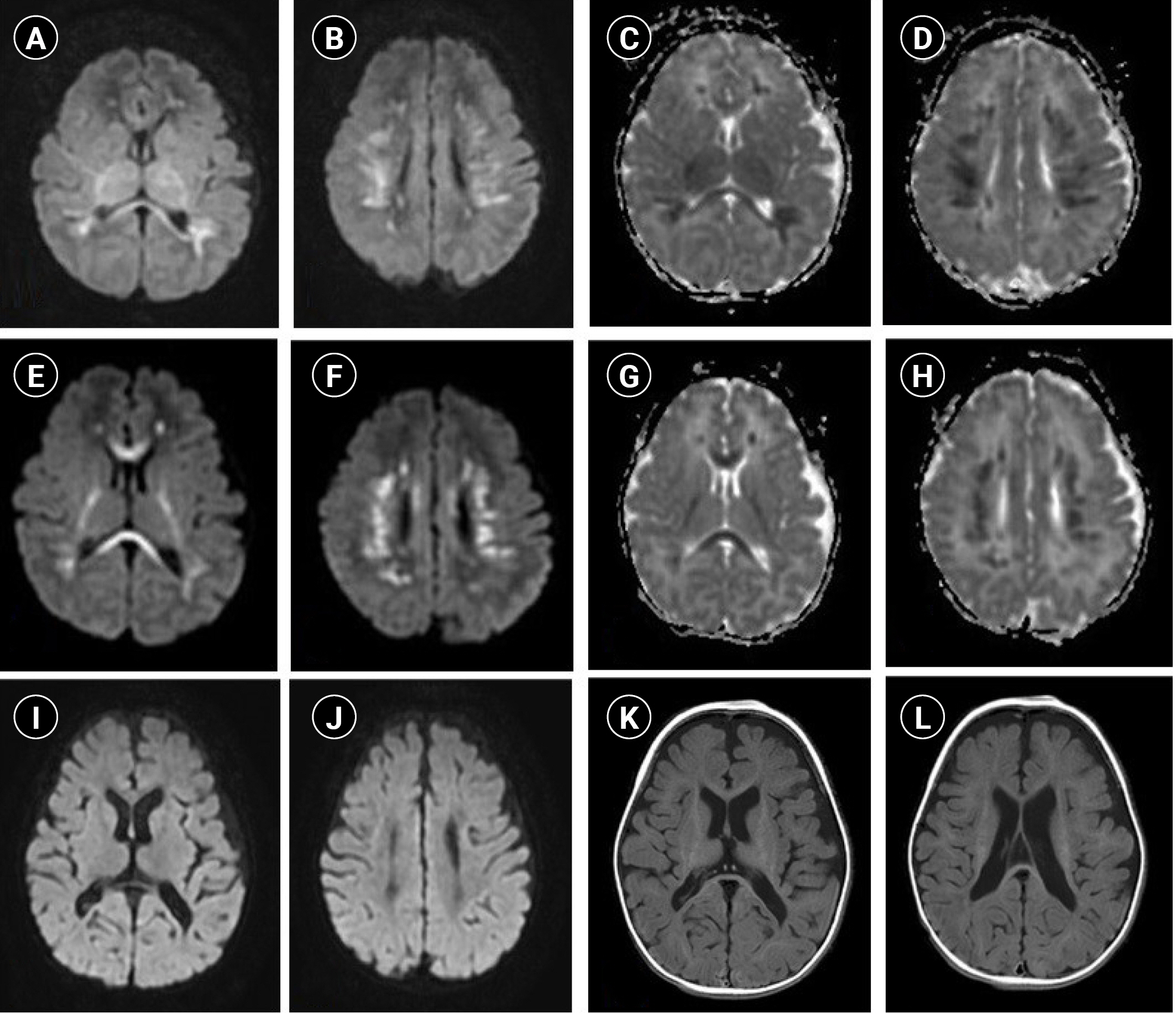
A 2-day-old girl was admitted for refractory hypoglycemia. The patient was born to a mother with gestational diabetes and hypothyroidism at the gestational age of 37 weeks and 6 days and had a birth weight of 2,460 g. After an uneventful delivery at a local hospital, her blood glucose level was 29 mg/dL. Despite glucose administration, the blood glucose level remained low (range, 23 to 34 mg/dL). She was subsequently transferred to our hospital. On admission, she was alert; however, she showed mild chest retraction with oxygen saturation of 90% under an oxygen supply of 1 L/min. Her heart rate, respiratory rate, body temperature, and blood pressure were 147 beats/min (range, 120 to 160), 52 breaths/min (range, 40 to 60), 36.7Ōäā (range, 36.5Ōäā to 37.4Ōäā), and 80/31 mm Hg (range, 60 to 90/30 to 60), respectively. Mechanical ventilation was applied to relieve her respiratory distress symptoms. The initial laboratory findings were as follows: serum glucose, 17 mg/dL; pH, 7.23; pCO2, 56.2 mm Hg; and bicarbonate, 23.6 mmol/L. The following were the results of the metabolic evaluation for hypoglycemia: insulin, 13.7 ╬╝IU/mL (range, 2.6 to 24.9); insulin/glucose ratio 0.5 (simultaneous serum glucose, 25 mg/dL, <0.3); free fatty acids, 93 ╬╝Eq/L (range, 172 to 586); ketone bodies, 0.2 mmol/L (<0.6); and cortisol, 9.13 ╬╝g/dL (range, 2.47 to 11.9). Even after intravenous glucose administration, her glucose level remained low, with a range of 31 to 52 mg/dL. Her glucose level stabilized following the administration of glucose infusion at a rate of 14 mg/kg/min and hydrocortisone. On the 4th day of admission, she developed multiple seizures with apnea, eyelid blinking, and lip-smacking under euglycemia with the same glucose management. Cerebrospinal fluid analysis revealed 2 white blood cells/╬╝L, a protein level of 81.8 mg/dL, and a glucose level of 68 mg/dL with a serum glucose level of 93 mg/dL. Immediately after the occurrence of seizures, we performed brain MRI on the 4th day of admission. Brain DWI and an apparent diffusion coefficient map demonstrated diffuse restricted water diffusion in the white matter of the parieto-occipital lobes, centrum semiovale, posterior limb of the internal capsule, and the splenium of the corpus callosum. On the 11th day, follow-up MRI showed the spread of lesions in the corpus callosum and internal capsule (Fig. 1). A stool test for rotavirus antigen was negative. Electroencephalography (EEG) showed a burst-suppression pattern on the 5th day of admission (Fig. 2). Intravenous anticonvulsants were administered to control seizures. She had five additional seizures after the first episode. Except for the first one, the others were subtle seizures with autonomic changes that comprised a sudden increase in heart rate up to 180 to 210 beats/min with decreased responsiveness to noxious stimuli for seconds. After the 12th day of admission, the seizures were controlled with medications (phenobarbital, 6 mg/kg/day; levetiracetam, 30 mg/kg/day). Follow-up EEG on the 12th and 15th days of admission showed an improving status of background activity despite persistent multifocal spikes and sharp waves. Considering the burst-suppression pattern on EEG and the topography of lesions, we performed whole-exome sequencing through GC Genome (Yongin, Korea) for the molecular diagnosis of possible white matter disorders, including other metabolic disorders causing hypoglycemia. Three heterozygous variants of unknown significance were revealed in sodium voltage-gated channel alpha subunit 1 (SCN1A) (NM_001165963.3:c.4096G>A;p.Val1366Ile), calcium voltage-gated channel subunit alpha1 H (CACNA1H) (NM_021098.3:c.3635C>T;p.Thr1212Ile), and MAF bZIP transcription factor A (MAFA) (NM_201589.4:c.738C>T;p.Gly246=).
Parental genetic testing revealed that the MAFA variant was observed in her mother, and the SCN1A and CACNA1H variants in her father. Family history revealed her mother and elder brother had a history of febrile seizures. On the 26th day of admission, she was discharged with medications and instructions for follow-up. At 7 months of age, brain MRI showed mild ventriculomegaly with an outer bulging contour, suggesting end-stage periventricular leukomalacia (Fig. 1). Over 14 months of follow-up, the patient had mild developmental delay with esotropia and hypertropia of the left eye. On the same follow-up day, her height was 76.2 cm (50th percentile), and her body weight was 9.3 kg (50th percentile).
MRI studies in patients with symptomatic hypoglycemia showed a correlation between the pattern of damage and age at clinical presentation: parieto-occipital white matter lesions from the neonatal period to 6 months of age versus basal ganglia and parieto-temporal cortex involvement in older infants [3]. To date, it remains unclear why the posterior head is more vulnerable to hypoglycemic injury in the neonatal period. The reduction of regional cerebral glucose use or a local expression deficit of glucose membrane transporter proteins and active synaptogenesis and axonal migration in the occipital lobe have been suggested as possible contributors to the localization of damage [4]. Burns et al. [2] studied the pattern of injury on early MRI scans following symptomatic hypoglycemia in 35 term infants. They found that the pattern of brain injury in symptomatic neonatal hypoglycemia was not associated with the severity or duration of hypoglycemia. Regarding the occurrence of seizures, all patients with seizures had white matter injuries; even four of the five patients without seizures had white matter injuries with variable degrees from mild to moderate [2]. According to other studies, severe or repetitive hypoglycemic brain injury is more often present with atypical MRI, and the sites of brain injury are numerous and extensive [5]. Diffuse, symmetric diffusion-restricted lesions involving the cerebral white matter can be observed in patients with neonatal seizures without any structural or metabolic etiology and in neonates with viral infections, such as rotavirus, human parechovirus, and enterovirus [6]. Our case had hypoglycemia for 3 days with the lowest level of 17 mg/dL. The patient developed multiple seizures for 9 days under euglycemia; however, she never developed status epilepticus. Her stool rotavirus test was negative. Based on the previous reports, the atypical MRI features of our case may have resulted from various factors, including the duration and severity of hypoglycemia, along with the presence of multiple seizures.
Although hypoxia-ischemia and hypoglycemia can each have different MRI patterns of injury, they can exert a combined negative effect on the developing brain. Mild to moderate hypoxic-ischemic injury in term neonates produces parasagittal watershed zone infarcts between the anterior/middle and the middle/posterior cerebral arteries [7]. Internal watershed strokes involve structures in the deep white matter that lie between medullary arteries arising from the superficial pial plexus and deep penetrating arteries arising from the basal cerebral arteries. These lesions lie in the corona radiata and centrum semiovale adjacent to the lateral ventricles [8]. In a study of 179 term infants, the watershed pattern of hypoxic-ischemic injury was most frequently observed to be associated with severe hypoglycemia [9]. When transferred, our patient showed respiratory distress as a possible clinical manifestation of hypoglycemia. Although our case had white matter lesions involving internal watershed zones that were not typical parasagittal areas, the MRI findings shown in our case may be an overlapping pattern of hypoglycemia and hypoxic-ischemic injuries.
Owing to atypical MRI features compared to the typical posterior dominant pattern of NHE, we performed an extensive workup in our patient, including whole-exome sequencing, which showed non-specific findings. Bilateral symmetric white matter changes are typically associated with toxic, metabolic, or hereditary leukoencephalopathies. NHE can present atypical MRI patterns differing from those predominantly focused in the posterior area with or without accompanying hypoxic-ischemic injury.
Our case showed unfamiliar findings caused by hypoglycemia. This additional information related to NHE could help physicians in medical practice.
Notes
Author contribution
Conceptualization: WJS and KHL. Data curation: WJS, YK, and KHL. Formal analysis: WJS, YK, and KHL. Methodology: WJS and YK. Project administration: KHL. Visualization: WJS, YK, and KHL. Writing-original draft: WJS. Writing-review & editing: WJS and KHL.
Fig.┬Ā1.
Serial brain magnetic resonance imaging (MRI) shows the evolution of white matter lesions in a neonate with symptomatic hypoglycemia from seizure onset to 7 months of age. Axial diffusion-weighted imaging (DWI) (A, B) and apparent diffusion coefficient (ADC) map (C, D) reveal restricted water diffusion in the white matter of the parieto-occipital lobes, centrum semiovale, posterior limb of the internal capsule, and the splenium of the corpus callosum on the 4th day of admission. Follow-up DWI (E, F) and ADC map (G, H) on the 11th day show no interval changes except for the spread of lesions in the corpus callosum and internal capsule. Follow-up MRI at 7 months of age shows mild ventriculomegaly with an outer bulging contour on T1-weighted imaging (K, L), with the disappearance of restriction of water diffusion observed in the previous images (I, J).
References
1. Poretti A, Blaser SI, Lequin MH, Fatemi A, Meoded A, Northington FJ, et al. Neonatal neuroimaging findings in inborn errors of metabolism. J Magn Reson Imaging 2013;37:294-312.



2. Burns CM, Rutherford MA, Boardman JP, Cowan FM. Patterns of cerebral injury and neurodevelopmental outcomes after symptomatic neonatal hypoglycemia. Pediatrics 2008;122:65-74.



3. Gataullina S, De Lonlay P, Dellatolas G, Valayannapoulos V, Napuri S, Damaj L, et al. Topography of brain damage in metabolic hypoglycaemia is determined by age at which hypoglycaemia occurred. Dev Med Child Neurol 2013;55:162-6.


4. Filan PM, Inder TE, Cameron FJ, Kean MJ, Hunt RW. Neonatal hypoglycemia and occipital cerebral injury. J Pediatr 2006;148:552-5.


5. Wang Y, Liu K, Ye X, Lu Y, Zhang X, Zhang C, et al. Typical and non-typical magnetic resonance imaging findings of neonatal hypoglycemic encephalopathy. Zhonghua Yi Xue Za Zhi 2014;94:2938-40.

6. Kim M, Hwang JY, Lee YJ, Kim YW, Byun SY, Lee YJ, et al. Clinical and imaging findings of neonatal seizures presenting as diffuse cerebral white matter abnormality on diffusion-weighted imaging without any structural or metabolic etiology. Taehan Yongsang Uihakhoe Chi 2020;81:1412-23.




7. Bano S, Chaudhary V, Garga UC. Neonatal hypoxic-ischemic encephalopathy: a radiological review. J Pediatr Neurosci 2017;12:1-6.



- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 3,110 View
- 82 Download
- Related articles in Ann Child Neurol
-
A Case of Spinal Muscular Atrophy with Extensive Involvement of Sensory Nerves.2006 November;14(2)
A Case of Brainstem Encephalitis Associated with Epstein-Barr Virus Infection.2011 December;19(3)