Anti-GD1b-Positive Miller Fisher Syndrome Presenting as Total Ophthalmoplegia
Article information
Miller Fisher syndrome (MFS) is a variant of Guillain-Barre syndrome (GBS) characterized by ataxia, areflexia, and ophthalmoplegia. It is often linked to cross-reacting antibodies to the GQ1b ganglioside found in cranial nerve myelin [1]. The presence of GQ1b at significantly higher levels in the oculomotor, trochlear, and abducens nerve myelin is often cited as an explanation for ophthalmoplegia [1]. Anti-GQ1b immunoglobulin G (IgG) antibodies are detected in more than 90% of patients with MFS [2]. However, we observed a case of MFS with severe ophthalmoplegia presenting as frozen eyes, in which anti-GQ1b IgG was negative, while anti-GD1b IgG was positive.
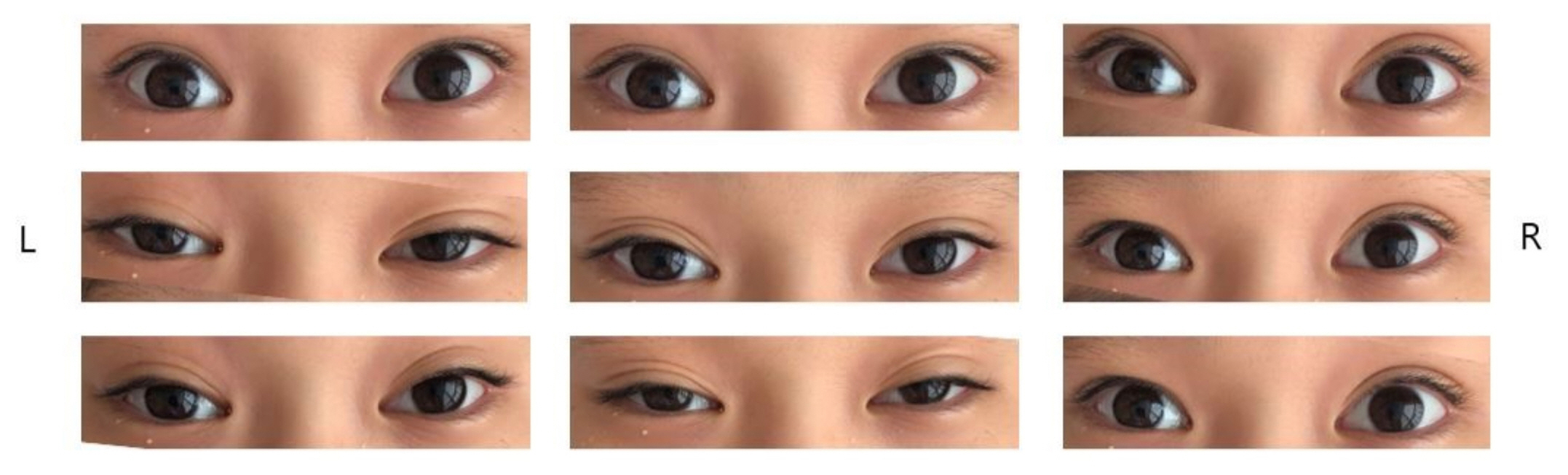
An 11-year-old boy presented to the outpatient clinic with headache, dizziness, vomiting, diplopia, ataxia, and a tingling sensation in the fingers for 3 days. He could not stand or walk without support. There was no fever at the time of presentation; however, 3 weeks ago he had experienced a high fever with loose stool for 2 days. His mental status was alert and his vital signs were within the normal range. His muscle strength was grade V in both the upper and lower extremities. The cranial nerve test was intact except for bilateral total ophthalmoplegia (Fig. 1). There was no nystagmus. Deep tendon reflex testing showed areflexia of both knees. On admission, brain magnetic resonance imaging (MRI) and whole-spine MRI were normal. Nerve conduction studies of both upper and lower extremities suggested acute inflammatory demyelinating polyneuropathy because both H-reflexes were absent. No electrophysiological abnormalities were observed in the left and right blink reflex pathways. On a blood test, the white blood cell count was 10,290/mm3 with 66.4% of segment neutrophils. The C-reactive protein level was 0.1 mg/dL. The thyroid function test was normal. Cerebrospinal fluid (CSF) examinations showed a white blood cell count of 2/mm3, a protein level of 25.5 mg/dL, and a glucose level of 62 mg/dL. Bacterial culture, herpes simplex virus polymerase chain reaction (PCR), and acid-fast bacillus stain of CSF were all negative. In a stool PCR examination, Campylobacter species were identified. Serum anti-GM1 IgG and anti-GQ1b IgG were negative, but anti-GD1b IgG was positive.
We administered intravenous immunoglobulin (0.5 g/kg/day) for 4 days upon admission under the diagnosis of MFS. The patient’s ataxia and headache immediately improved after treatment. However, only adduction of the left eye was possible immediately after the treatment. The tingling sensation of the fingers improved after 2 weeks. Ophthalmoplegia slowly improved. Two months later, bilateral total ophthalmoplegia had completely improved.
Total bilateral ophthalmoplegia is very rare. Various combinations of internal and external ophthalmoplegia are generally reported in anti-GQ1b antibody syndromes, including acute ophthalmoplegia without ataxia, MFS, GBS with ophthalmoplegia, MFS/GBS overlap syndrome, and Bickerstaff’s brainstem encephalitis [3,4]. Rare cases of total bilateral ophthalmoplegia have been reported in MFS with anti-GQ1b antibody and Wernicke encephalopathy [5,6]. GD1b is densely localized on the dorsal root ganglia neurons, and antibodies to GD1b are known to cause ataxic neuropathy rather than ophthalmoplegia [7]. A previous study reported that patients with anti-GD1b antibodies of GBS and GBS/MFS tend to recover faster regarding the ability to walk [8]. The rapid recovery of walking in our case is in line with those results. Recently, in coronavirus disease 2019 (COVID-19)-associated MFS patients, antibodies against anti-GD1b instead of anti-GQ1b have been found [9]. As a proposed explanation, it was pointed out that the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike protein also binds to sialic acid-containing glycoproteins for cell entry, and anti-GD1b antibodies might exhibit cross-reactivity between SARS-CoV-2–bearing gangliosides and peripheral nerve glycolipids [9]. In our case, infection with Campylobacter species carrying GD1b-like lipooligosaccharides might have induced antibodies to GD1b, which caused MFS. We also consider the possibility of a cross-reaction between anti-GD1b antibodies and anti-GQ1b antibodies. However, a further evaluation would be needed to investigate additional factors causing severe ophthalmoplegia accompanying demyelination of the oculomotor, trochlear, and abducens nerves with negative anti-GQ1b antibodies.
This study was approved by the Institutional Review Board of Gachon University Gil Medical Center (GDIRB2021-430). Written informed consent was obtained from the guardian of the patient.
Notes
No potential conflict of interest relevant to this article was reported.
Author contribution
Conceptualization: HJK. Data curation: SJH. Formal analysis: HJK. Visualization: SJH. Writing-original draft: SJH. Writing-review & editing: HJK.