Screening Children with Epilepsy for Cognitive Deficits Using the Modified Mini-Mental Scale Examination and the Digit Letter Substitution Test
Article information
Abstract
Purpose
Children with epilepsy commonly have cognitive deficits; however, full-length neuropsychological testing is time- and resource-intensive. Therefore, we evaluated the feasibility of using the modified Mini-Mental Scale Examination (MMSE) and the Digit Letter Substitution Test (DLST) to screen children with epilepsy for cognitive deficits.
Methods
This was a prospective case-control study comparing scores on the MMSE and the DLST in children with epilepsy with normal age-matched controls between 8 and 12 years of age.
Results
In 35 cases and 36 controls, the cases had significantly lower (P<0.05) mean scores than the controls. The correlation coefficient between the MMSE and DLST scores was 0.902 (P<0.001). Children with developmental or speech delays and an epilepsy duration ≥5 years had lower scores than those without the corresponding risk factors.
Conclusion
This study demonstrated significantly lower scores on the MMSE and DLST in children with epilepsy than in controls, as well as significantly lower scores in patients with developmental or speech delays and an epilepsy duration ≥5 years.
Introduction
Cognitive deficits are seen in approximately half of patients with epilepsy, even at diagnosis [1]. A community-based cohort study of children with epilepsy demonstrated that 26.4% had subnormal global cognitive function [2]. Over time, while the majority of these children maintain their cognitive levels, children with uncontrolled seizures, especially younger children, have shown more than a 10-point drop in the full-scale intelligence quotient [3]. Analyses of the adult outcomes of childhood epilepsy have shown that those with good cognitive skills fare significantly better than their counterparts with poor cognitive skills in several domains, including education, employment, marriage and parenthood [4].
The etiology of epilepsy determines cognitive functioning to a large extent. However, seizures also interfere with learning and behaviour, especially in the developing brain, most likely by interfering with signalling pathways and neuronal networks [5]. The degree and progression of cognitive deficits in children with epilepsy are important to identify and monitor. Hence, the International League Against Epilepsy (ILAE) Neuropsychology Task Force recommends that all children with newly diagnosed epilepsy should receive routine screening for cognitive difficulties [6]. Detailed neuropsychological function testing, if done thoroughly, requires 6 to 8 hours of contact between the patient and examiner. Even a highly abbreviated assessment takes at least 2 hours [7]. While these comprehensive test batteries are the gold standard for evaluation, they are unavailable at most centres, especially in developing countries. To date, few studies have used screening tools to detect cognitive dysfunction in children with epilepsy. Even in the United States, a survey including 25 unique epilepsy programs showed that 50% of them used non-standardised informal questioning to screen for cognitive deficits [8]. There is an urgent need for a simple, quick, and repeatable screening tool that clinicians may use. Abnormal results on a screening tool could prompt a recommendation for detailed neuropsychological testing.
The first test we chose was the modified Mini-Mental Scale Examination (MMSE). The MMSE evaluates orientation, attention and concentration, registration, recall, and language and has been widely used as a short screening tool for cognitive impairment, especially in those above 4 years of age [9]. An evaluation of 181 Spanish children found that it correlated well with both the chronological age and Kaufman Brief Intelligence test [10]. A modification of the MMSE called School Years Screening Test for Evaluation of Mental Status (SYSTEMS) was tested in Australian school children and showed adequate sensitivity and specificity in detecting cognitive dysfunction [11]. We used the MMSE, which was standardised for Indian children and further modified for Brazilian children [12,13].
The broad subgroups of cognitive function that are commonly tested include intelligence, language, memory, executive function, and psychomotor speed. Of these subgroups, psychomotor processing speed has been shown to be the most affected in children with both localised and generalised epilepsy [14]. The Digit Letter Substitution Test (DLST) requires participants to quickly match various symbols such as letters with other symbols such as numbers in a limited time. It is one of the commonest tests in neuropsychology, and it is considered a good test for monitoring information processing speed in addition to a range of other cognitive functions including motor speed, attention, visuo-perceptual functions, visual scanning, and manual dexterity. The DLST has been shown to yield clinically meaningful results when serially monitoring patients [15]. Normative data for the DLST in Indian children are available; hence, this was the second test we chose to evaluate [16].
The present study aimed to evaluate the feasibility of using these two simple screening cognitive tests (the MMSE and DLST) in children with epilepsy. Each of these tests requires about 10 to 15 minutes to administer and score.
Materials and Methods
1. Patient selection
This was a prospective case-control study. The inclusion criteria for cases were (1) children with epilepsy; (2) age between 8 and 12 years; and (3) the ability to read and write letters and numbers. Cases were excluded if they refused consent or were unable to use paper and pencil due to motor deficits. Cases were selected among children with epilepsy attending the Pediatric Neurology Clinic of Choithram Hospital and Research Centre between July 2019 and June 2021. Controls included children between 8 and 12 years of age without neurological illnesses who attended the general pediatric outpatient department or were admitted to the pediatric ward for non-neurological illnesses.
2. Assessment of cases and controls
The following data were collected from cases: age, sex, significant perinatal events, age at onset of epilepsy, duration of epilepsy, etiology of epilepsy, type of seizures (focal or generalized), seizure burden, age of acquisition of developmental milestones, date of last seizure, electroencephalogram (EEG) reports, magnetic resonance imaging (MRI) brain reports, and the number of anti-epileptic drugs (AEDs). Data collected from controls included age, sex, age of acquisition of developmental milestones, and significant perinatal events.
3. Evaluation
All children were evaluated using the MMSE and the DLST. Testing was done in a quiet room after a careful explanation of the test to the child. The MMSE for children was calculated based on a set of questions and tasks given to the child, and their answers were scored based on a test previously standardised in Indian children [12]. It included five subtests to assess orientation, attention/concentration, registration, recall, and language. The cut-off scores for abnormal results were 28, 30, and 35 in the age groups of 6–8, 9–11, and 12–14 years, respectively [12].
The DLST is a paper-and-pencil cognitive test in which the patient is asked to match a set of digits with letters according to a key shown at the top of the page. The children were asked to match as many target digits as possible in the specified time of 90 seconds. The net score was obtained by deducting wrong substitutions from the total substitutions attempted. Scores below 2 standard deviations for age and sex were considered abnormal [16,17].
4. Ethics
The study was approved by the local institutional ethics committee (IER/2021/41). Informed consent was obtained from the parents or guardians accompanying the children.
5. Statistical analysis
The data were analysed using GraphPad and Epi Info as online-available software. Comparisons of means between the two groups were conducted using the unpaired t-test. The Glass delta value was used to calculate the effect size when the standard deviation of the study and control groups differed, and the Hedge g value was used if the sample sizes were different [18]. Correlations between pairs of parametric variables were calculated using the Pearson coefficient of correlation test. A P value of <0.05 was interpreted as statistically significant. Scores on the MMSE and DLST were compared in cases with and without various risk factors that have been described in literature as being associated with cognitive deficits, such as the age of onset of seizures below 2 years, a high seizure frequency, long duration of epilepsy, MRI brain abnormalities, and a relatively high number of AEDs.
Results
1. Baseline data
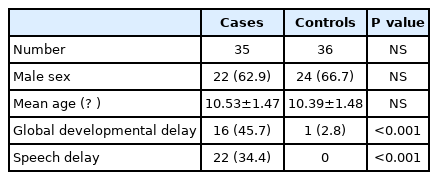
There were 35 cases (62.9% male) and 36 controls (66.7% male) (Table 1). The mean age of cases was 10.53±1.47 years, and that of controls was 10.39±1.48 years. Global developmental delay was seen in 16 (45.7%) cases and one (2.8%) control (P<0.001). Speech delay was seen in 22 (34.4%) cases and in none of the controls (P<0.001).
2. Details of cases
Generalised-onset epilepsy was seen in 23 (65.7%) cases, while 12 (34.3%) had focal onset epilepsy (Table 2). Significant perinatal events, including birth asphyxia and neonatal hypoglycemia, were noted in 16 patients. The etiology of epilepsy was idiopathic in 22 cases (62.9%), perinatal hypoxia in 10 (28.6%) and structural in three (8.6%). MRI brain abnormalities were seen in 20 patients (57.1%) and EEG abnormalities in 15 (42.9%). The age at onset of epilepsy was below 2 years in 15 (42.9%), between 2 and 5 years in 11 (31.4%), and above 5 years in nine (25.7%). The seizure burden was 0–5 per month in 20 patients (57.1%), 6–20 per month in 10 (28.6%), and more than 20 per month in 5 (14.3%). Ten children (28.6%) were taking one AED, 16 (45.7%) were taking two AEDs, six (17.1%) were taking three AEDs, and three (8.6%) were taking no AEDs. Fifteen (42.8%) children had experienced no seizures for the past 1 year or more and were classified as having controlled epilepsy.
3. Scores on cognitive screening tests in cases and controls
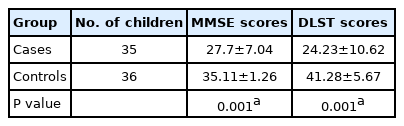
The mean scores of children with epilepsy were significantly lower (P<0.05) than those of controls on the MMSE (28 vs. 35) and DLST (24 vs. 41) (Table 3). Subnormal cognitive scores were seen in 21 (60%) and 13 (37%) of the case group using the MMSE, and DLST, respectively, while none of the children in the control group had subnormal scores on any of the tests.
4. Comparison of scores on the MMSE versus the DLST
The correlation coefficient between MMSE and DLST scores was 0.902 (P<0.001), demonstrating a very high correlation between the two tests.
5. Comparison of MMSE and DLST scores in children with epilepsy with and without various risk factors for poor cognition
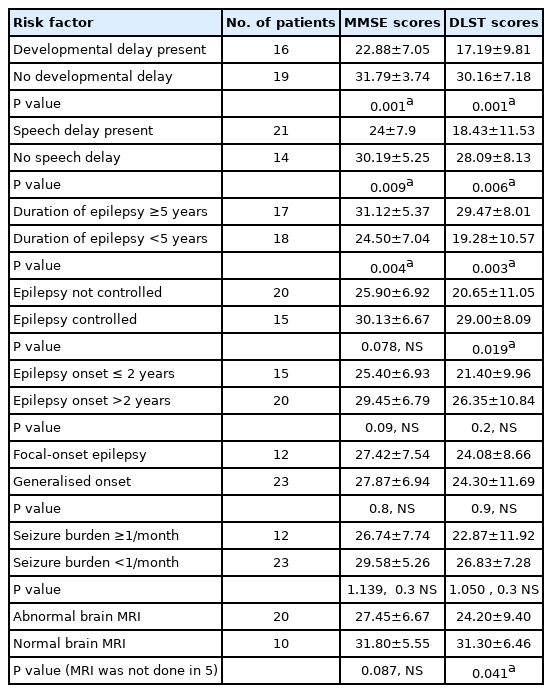
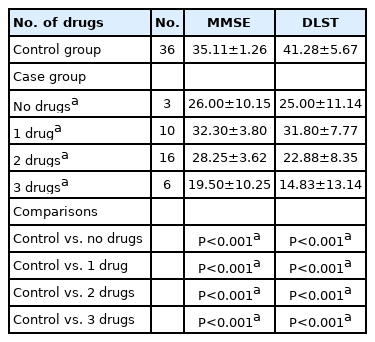
The MMSE and DLST scores in children with epilepsy with various risk factors for poor cognition were compared with those of children with epilepsy who did not have those risk factors. The risk factors included developmental delay, speech delay, duration of epilepsy ≥5 years, uncontrolled epilepsy (defined as the presence of seizures in the past 1 year), age of onset of epilepsy ≤2 years, focal-onset epilepsy, seizure burden ≥1/month, and abnormal brain MRI. Children with developmental delay, speech delay, and duration of epilepsy ≥5 years all showed significantly lower scores on both the MMSE and DLST than their counterparts without these risk factors (Table 4). Children with uncontrolled seizures and abnormal MRI findings showed significantly lower scores on the DLST, but not on the MMSE, as compared to children without these risk factors. However, children with an age of onset of epilepsy ≤2 years, focal-onset epilepsy and those with a high seizure burden did not show significantly lower scores than controls. All scores fell uniformly as the number of AEDs rose from 1 to 3. The mean scores of epileptic children on no drugs were lower than those on one or two drugs but higher than those on three drugs (probably because two patients were non-compliant and one had not begun anti-seizure medication) (Table 5).
Discussion
In this study, we found that it was feasible to use two previously validated quick tests for cognitive deficits (namely, the MMSE and the DLST) in a clinical setting for children with epilepsy. Children with epilepsy had significantly lower mean scores than normal age-matched controls on both the MMSE and DLST. The scores on both tests correlated well with each other. Children with several known risk factors for low cognition scored lower than those without the corresponding risk factors.
While the ILAE Neuropsychology Task Force recommends routine screening for cognitive deficits in children with epilepsy, they do not mention any particular screening test. Nonetheless, they suggest that testing should cover most core cognitive domains such as general intellect, attention, speed of processing, memory, language, spatial function, executive function, and sensory and motor function [6]. The MMSE has the advantage of screening most cognitive domains. However, it is more complicated to use than the DLST, which has the advantage of simplicity but evaluates a narrower window of cognitive functions, focusing primarily on processing speed. There is always a trade-off between what is practical versus what is ideal; however, the high concurrent validity between the tests suggests that the DLST may be useful in a busy clinical setting. The DLST has been used to serially monitor cognitive decline in children with multiple sclerosis. An increase in DLST scores by 4 points or 10% is now being used to identify and define clinical responders in these patients [19].
There are few reports of the use of cognitive screening in children with epilepsy. Triplett and Asato [20] studied 39 children with medication-naive epilepsy between 8 and 17 years of age using the computerised Central Nervous System (CNS) Vital Signs battery, which is a 30-minute assessment over multiple cognitive domains. Their pilot study found that this battery was time-efficient, potentially usable in the clinical setting, and well tolerated by children. However, this computerised battery needs to be purchased and may not be accessible to clinicians working in developing countries. Both tests we used are brief and take 10 to 15 minutes to administer and score. Furthermore, they are freely available and need no additional infrastructure other than paper and pen. They have been validated in chil-dren of various age groups. While computerised testing has advantages in terms of the standardisa-tion and accuracy of timing, it has inherent problems. Some limitations include its availability in low-resource countries and variation in stimuli due to differences in screen resolution. Moreover, much clinical information is garnered by observing the patient’s interaction styles and the way a patient responds to difficulties, but those nuances are lost in computerised testing. The six-item PedsQL (https://www.pedsql.org/) cognitive functioning scale has recently been used as a brief generic outcome measure in children with epilepsy, but this is a patient-/parent-reported self-assessment scale [21].
Although the main goal of this study was to assess the feasibility of the use of two simple screening tests for cognitive deficits in children, we also explored risk factors for low scores to support construct validity. In the literature, risk factors for cognitive deficits include epilepsy onset in early childhood, uncontrolled epilepsy, symptomatic epilepsy, a higher seizure burden, poor seizure control, and relatively high AED exposure [3,22-25]. In our study, children with global developmental delay, delayed speech, and epilepsy duration ≥5 years all had significantly lower scores on the screening tests than the normal controls. However, in children with uncontrolled epilepsy and those with abnormal brain MRI, the scores of cases were lower than those of controls only in the DLST, while the MMSE failed to show a statistically significant difference. In contrast, seizure onset at ≤2 years of age, generalised epilepsy, and a seizure burden ≥1/month were not associated with significantly lower scores than controls on both tests. There is a clear need for studies including larger groups of children.
The lack of simultaneous testing of all children with a gold-standard test for cognition such as the Wechsler Full-Scale Intelligence Quotient and the relatively small sample size are the major limitations of the study. The study was carried out among children with epilepsy referred to a tertiary care centre, who therefore constituted a group with significant cognitive issues. It would be relevant to subsequently evaluate these tests in a larger group of children with epilepsy who have no apparent developmental or cognitive delays to evaluate its efficacy in identifying and monitoring children with subtle deficiencies who would benefit from a detailed evaluation and intervention.
In summary, this pilot study evaluated the feasibility of using the MMSE and the DLST to screen children for cognitive deficits. We found the two tests easy to use, and their results were closely correlated with each other. These tests appear potentially useful to screen and monitor for cognitive deficits in children with epilepsy. Further comparisons with gold-standard full-length neuropsychological tests on larger groups of children with epilepsy will clarify their value and help us identify children at risk and intervene in time.
Notes
No potential conflict of interest relevant to this article was reported.
Author contribution
Conceptualization: GRP. Data curation: MB. Formal analysis: GRP. Methodology: MB and GRP. Project administration: GRP. Visualization: GRP. Writing-original draft: GRP. Writing-review & editing: GRP.