 |
 |
- Search
| Ann Child Neurol > Volume 27(1); 2019 > Article |
|
Abstract
In this article, we reviewed current knowledge regarding gene-specific therapies for some developmental and epileptic encephalopathy caused by genes with high diagnostic yields, and which are therefore, also more frequently encountered by physicians during treatment, including ALDH7A1, CDKL5, KCNQ2, KCNT1, SCN2A, SCN8A, STXBP1, and SYNGAP1. Among these therapies, the ones directly targeting causative mutations are retigabine in KCNQ2 encephalopathy and quinidine in KCNT1 encephalopathy. However, despite promising results in vitro, the outcomes related to these therapies were disappointing when administered to patients. Considering the pathologic mechanisms of causative mutations, sodium channel blockers are recommended for patients with KCNQ2 mutations, infantile epileptic encephalopathy patients with SCN2A mutations, and patients with SCN8A mutations. Levetiracetam can be considered for patients with STXBP1 mutations.
With the advent of next-generation sequencing, there have been significant advances in the genetics of epilepsy within the last decade [1]. In developmental and epileptic encephalopathy patients, targeted gene panels and/or whole exome sequencing have now become part of the routine diagnostic workup, which provide a genetic diagnosis in about 30% of patients.
As the number of genetically diagnosed patients has increased, so has the demand for more precise treatment based on pathogenic genetic mutations. The demand has resulted in active research and a rapidly increasing number of publications focused on precision medicine in epilepsy (Fig. 1) [2]. Based on current knowledge, approximately 25% of genetically diagnosed epilepsy patients with de novo monogenic mutations carry potential targets for precision medicine approaches [3].
In this article, we review current knowledge regarding gene-specific therapies for some forms of developmental and epileptic encephalopathy caused by genes with high diagnostic yields, which are therefore, more frequently encountered by treating physicians.
The aldehyde dehydrogenase 7 family, member A1 (ALDH7A1) gene encodes the enzyme ╬▒-aminoadipic semialdehyde (╬▒-AASA) dehydrogenase, also known as antiquitin, a key enzyme in lysine metabolism [4]. Biallelic mutations of ALDH7A1 result in deficiency of ╬▒-AASA dehydrogenase, which then results in the accumulation of ╬▒-AASA and the cyclic equivalent ╬ö-1-piperideine-6 carboxylate (╬ö1-P6C) [4]. The accumulated ╬ö1-P6C sequesters the active form of vitamin B6 (pyridoxal 5ŌĆ▓-phosphate [PLP]), and causes pyridoxine-dependent epilepsy [4].
The classic presentation of pyridoxine-dependent epilepsy is the neonatal onset of treatment-resistant seizures that respond dramatically, both clinically and electroencephalographically, to pyridoxine supplementation [5]. Despite adequate seizure control achieved with pyridoxine alone, 75% of patients with pyridoxine-dependent epilepsy have significant intellectual disability and/or developmental delay [6]. The degree of intellectual disability or developmental delay does not correlate with seizure control or the age at which pyridoxine treatment is initiated [6].
Treatment with pyridoxine supplementation in pyridoxine-dependent epilepsy patients is therapeutic because it overcomes the secondary depletion of pyridoxal phosphate (PLP) [5]. Pyridoxine supplementation alone does not treat the underlying defect of lysine oxidation, but treatment with a lysine-restricted diet and/or with a lysine transport inhibitor (arginine) has been shown to improve cognitive function at some level [7-9]. Triple therapy with pyridoxine, arginine supplementation, and dietary lysine restriction has been recommended to ameliorate the cognitive impairment seen in pyridoxine-dependent epilepsy [9].
Cyclin-dependent kinase-like 5 (CDKL5) encephalopathy is a severe X-linked neurodevelopmental disease caused by mutations in the CDKL5 gene, which lead to deficiency in CDKL5 protein expression or function [10]. CDKL5 is highly expressed in the brain, mainly in neurons, with both nuclear and dendritic localization, and its expression peaks during the postnatal period [11,12]. In mouse models, Cdlk5 knockout animals exhibited hippocampus-dependent learning and memory impairment, visual and respiratory deficits, and motor stereotypes, which were associated with neuroanatomical alterations such as reduced dendritic branching of hippocampal and cortical neurons, reduced dendritic spine density, and altered connectivity [13-19].
Epilepsy in CDKL5 encephalopathy patients often starts before 2 months of age, with frequent generalized tonic seizures that typically last for less than 1 minute [20]. Seizures at this time are well-controlled with antiepileptic drugs (AEDs), and electroencephalograms (EEGs) are usually normal [20]. Epilepsy relapses in late infancy at a median age of 11 months, when infantile spasms with hypsarrhythmia, sometimes combined with brief tonic medication-resistant seizures, emerge [20]. During the year after onset of infantile spasms, epilepsy evolves into late multifocal and myoclonic epilepsy, showing frequent pharmacoresistant seizures of multiple types such as myoclonic seizures, and EEGs showing high-amplitude delta waves with pseudo-periodic bursts of high-amplitude spikes [20]. Severe neurodevelopmental delay is present from the beginning, but there is usually no period of regression [21].
Seizures in CDKL5 encephalopathy, especially seizures during the ŌĆ£late epilepsyŌĆØ stage are highly refractory, and there are no available AEDs that are effective in controlling these seizures. A promising study regarding cannabidiol came out in 2018, which analyzed patients with treatment-resistant, childhood-onset epilepsies including CDKL5 encephalopathy [22]. In CDKL5 encephalopathy patients (n=17), 50% responder rate was 41% at week 12, and 53% by week 48 [22]. However, cautious interpretation is needed due to the small number of subjects and due to exclusion of withdrawn patients from analysis. Therefore, additional placebo-controlled randomized trials with larger sample sizes are needed.
Several labs are assessing the use of drugs such as tianeptine and tideglusib in CDKL5 encephalopathy. Tianeptine is an antidepressant that has been used for more than 30 years. Studies utilizing neurons from Cdkl5 knockout mouse models have proposed that CDKL5 deficiency in primary hippocampal neurons negatively affects the expression of the alpha-amino-3-hydroxy-5-methyl-4-iso-xazole propionic acid (AMPA) receptors, and such an effect is likely to contribute, at least in part, to the altered synaptic functions and cognitive impairment linked to loss of CDKL5 [23]. Therefore, tianeptine, which is known to recruit and stabilize AMPA receptors at the synaptic sites, has been studied in vitro and results revealed that tianeptine normalized the expression of membrane-inserted AMPA receptors [23].
Tideglusib is a glycogen synthase kinase 3 beta (GSK3╬▓) inhibitor, and is currently undergoing clinical trials for use against diseases such as AlzheimerŌĆÖs, progressive supranuclear palsy, and myotonic dystrophy. In mouse, deficiency of Cdkl5 caused defects in postnatal hippocampal development and hippocampus-dependent learning and memory. These defects were accompanied by increased activity of GSK3╬▓, an important inhibitory regulator of many neuronal functions [17]. Therefore, tideglusib was tested on Cdkl5 knockout mice and results showed that tideglusib improved hippocampal development, hippocampus-dependent behaviors, and memory performance in juvenile Cdkl5 knockout mice [24].
Finally, CDKL5 protein substitution therapy was successfully conducted in a mouse model using a protein transduction domain (TAT) that was able to deliver macromolecules into cells, and even into the brain when fused to a specific protein [25]. Intracerebroventricular infusion of TAT-CDKL5 protein was efficiently internalized by target cells, which resulted in the retention of CDKL5 activity and the restoration of hippocampal development, hippocampus-dependent memory, and breathing pattern in Cdkl5-null mice [25]. Systemically administered TAT-CDKL5 protein also crossed the blood-brain barrier, reached the central nervous system, and improved behavioral defects in mice [25].
Mutations in the potassium channel, voltage-gated, KQT-like subfamily member 2 (KCNQ2) gene, encoding voltage-gated K+ channel subunits underlying the neuronal M-current, usually lead to loss-of-function of KV7.2 and cause a decrease in neuronal M-current conductance, thereby increasing neuronal excitability [26]. Functional studies have revealed that a 25% reduction in KV7.2 current is sufficient to increase neuronal excitability to epileptogenic levels in early infancy [27]. However in rare cases, variants of KCNQ2 such as R144Q, R198Q, R201C, and R201H, produce gain-of-function effects and are reported to be associated with early-onset epileptic encephalopathy, infantile spasms, or neonatal nonepileptic myoclonus [28-30].
Benign familial seizures of early infancy and early infantile epileptic encephalopathy (EIEE) are two main types of epilepsy associated with KCNQ2 mutations. Seizure onset in both syndromes occurs as early as in the first days of life [31]. Patients with benign seizures in early infancy have an excellent prognosis regarding both seizure remission and neurodevelopment. In contrast, patients with EIEE suffer from a severe phenotype comprising drug-resistant seizures, intellectual disability, and encephalopathic EEG [31].
A systematic review conducted in 2019 analyzed the medications used in EIEE, and revealed that sodium channel blockers were the most frequently used monotherapy agent [31]. The distribution of AEDs used by patients achieving control of their seizures was 37.8% with sodium channel blockers, 26.3% with valproic acid, and 33.3% with levetiracetam [31]. Treatment trials with phenobarbital were largely unsuccessful: 8.6% of the trials led to seizure freedom in patients while 74.3% of the trials showed no effect [31]. Therapeutic response to sodium-channel blockers in patients with KCNQ2 mutations could be explained by the fact that voltage-gated sodium channels and KCNQ potassium channels co-localize at critical locations on the neuronal membrane and therefore, modulation of one channel may significantly affect the function of the channel complex [32,33]. Because of this, sodium channel blockers such as phenytoin, carbamazepine, and oxcarbazepine are recommended as a first-line treatment [31,34]. However, sodium channel blockers were not clearly superior to levetiracetam or valproate, indicating that the latter AEDs might be considered in patients who failed to respond to sodium channel blockers [31].
Retigabine (also known as ezogabine) is a Kv7.2/Kv7.3 opener that leads to neuronal hyperpolarization of the membrane potential and was first introduced as an adjunctive therapy in adults with focal seizures [35]. Despite its effectiveness, retigabine was withdrawn from the market due to lack of demand caused mainly by side effects such as blue discoloration of skin and retina [36]. However, the fact that retigabine opens the Kv7 potassium channel drew interest in its use for patients with KCNQ2 mutations, and the results were favorable in animal models [37]. Of all mono-therapeutic treatment trials with retigabine, only 14.3% led to seizure freedom while 71.4% showed no effect and current findings do not support use of retigabine compared to other drugs such as sodium channel blockers [31].
The potassium channel subfamily T member 1 (KCNT1) gene encodes the sodium-activated potassium channel KCa4.1. KCNT1 is widely expressed throughout the brain, as well as in the dorsal root ganglia, kidney, and heart, and is responsible for slow hyperpolarization after bursts of action potentials [38,39]. The pathogenic KCNT1 mutations described to date are all missense variants; no nonsense or other truncating mutations have been identified yet [40]. This suggests that perturbation of normal KCNT1 protein function, rather than the loss of its function, is the underlying pathomechanism [40]. This is consistent with other reports, including animal models demonstrating that KCNT1 pathogenic variants manifest a gain-of-function effect with increased current amplitude [39,41,42].
Mutations in KCNT1 cause a wide spectrum of epileptic disorders with variable ages at onset and cognitive outcomes including Ohtahara syndrome, West syndrome, and severe nocturnal frontal lobe epilepsy (NFLE). However, epilepsy of infancy with migrating focal seizures (EIMFS) and autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE) are the two most characteristic epilepsies associated with KCNT1 [39,41,43,44]. KCNT1 is the most frequently identified genetic cause of EIMFS, and 39% (28 out of 71) of individuals from EIMFS patient cohorts that have been analyzed for KCNT1 mutations are mutation positive [40]. Patients with ADNFLE and NFLE with KCNT1 mutations have a more severe phenotype than ADNFLE patients with mutations in nicotinic acetylcholine receptor subunit genes in terms of earlier age of seizure onset, marked increase in frequency of refractory seizures, and high frequency of comorbid intellectual disability and psychiatric features [44].
Quinidine is an inhibitor of several types of potassium channels, including KCNT1, and is currently used as an antiarrhythmic agent and antimalarial drug [40,45,46]. Given that functional studies have shown mutations in KCNT1 cause an increase in channel current, quinidine gathered interest as a potential inhibitor that could reverse this abnormal channel function and therefore, treat seizures in patients with mutations in KCNT1 [41,42,47,48]. Indeed, the effectiveness of quinidine in reversing the increased current of mutant KCNT1 channels has been demonstrated in vitro [42,48]. However, in an order-randomized, blinded, placebo-controlled, crossover trial of quinidine in six ADNFLE patients, 33.3% (n=2) of patients discontinued therapy due to prolonged QT interval occurring at serum quinidine levels below the therapeutic range, and none of four patients who completed treatment trial showed 50% seizure reduction [49]. Other reports that included 25 EIMFS, two focal epilepsy, one West syndrome, and one NFLE patients, demonstrated that 31.0% (n=9, including eight EIMFS) of patients experienced improvements in seizure activity due to quinidine administration while the others did not show any improvements [39,48,50-60]. Lack of therapeutic response observed in the majority of patients is likely because exposure levels in the brain are too low to cause significant in vivo channel blockade [52].
The sodium channel, voltage-gated, type II, alpha subunit (SCN2A) gene encodes the neuronal sodium channel NaV1.2, one of four sodium channel paralogs expressed throughout the central nervous system, along with NaV1.1 (SCN1A), NaV1.3 (SCN3A), and NaV1.6 (sodium channel, voltage-gated, type VIII alpha subunit [SCN8A]) [61]. Mutations in SCN2A are primarily associated with three distinct disorders: (1) infantile epileptic encephalopathy characterized by drug-resistant seizures with age of onset less than 12 months, followed by neurodevelopmental delay; (2) benign (familial) infantile seizures characterized by seizure onset at less than 12 months of age, which resolve by 2 years of age without overt long-term neuropsychiatric consequences; and (3) autistic spectrum disorder/intellectual disability (ASD/ID) characterized by global developmental delay, particularly of social and language milestones, with up to one-third of children developing childhood-onset seizures after 12 months of age [61-63].
An integrated analysis of genetic and electrophysiological data suggested a model explaining the three disorders associated with SCN2A (Fig. 2) [61]. Variants associated with gain-of-function of NaV1.2 channel activity led to infantile epileptic encephalopathy and benign infantile seizures, while those associated with loss-of-function led to ASD/ID [62,63]. Further, the degree to which the gain-of-function variants potentiate NaV1.2 activity distinguishes infantile epileptic encephalopathy from benign infantile seizures, with severe variants leading to infantile epileptic encephalopathy [62,63]. This model is supported by several other findings including observations using in vitro electrophysiology, the restriction of protein truncating variants resulting in loss of function in ASD/ID cases, shared symptoms between individuals with recurrent missense variants, and the clustering of infantile epileptic encephalopathy/benign infantile seizure variants around the voltage sensor domain of the channel while the ASD/ID missense variants cluster near the pore loop regions [62,63].
Seizures in SCN2A encephalopathy are often resistant to AEDs. However, for infants less than 4 months of age, sodium channel blockers such as phenytoin and carbamazepine are more effective, which is expected considering the gain-of-function nature of causative mutations [62,63]. For children more than 12 months of age with ASD/ID and childhood-onset seizures, the opposite action should be taken, and drugs other than sodium channel blockers, such as levetiracetam, benzodiazepines, and valproate, are therapeutic options. New emerging therapies such as bromides, fenfluramine, and cannabidiol are being studied and have shown efficacy in treating Dravet syndrome [64]. Whether such medications would provide benefit with SCN2A associated late-onset seizures remains unknown. However, when considering such treatments, it is important to remember that the NaV1.1 channels affected in Dravet syndrome are more commonly expressed in inhibitory neurons, and that loss-of-function of NaV1.1 may have opposing effects in brain networks compared with NaV1.2 loss-of-function in excitatory neurons [65].
The SCN8A gene encodes the pore-forming voltage-gated sodium channel subunit Nav1.6, which is widely expressed in the brain and is responsible for the initiation and propagation of neuronal action potentials contributing to regulation of neuronal excitability [66-68]. Most mutations identified in SCN8A result in gain-of-function changes in biophysical properties leading to elevated channel activity, either due to premature channel opening or impaired channel inactivation [69-73]. However, a small subset of mutations cause loss of function of SCN8A, resulting in isolated intellectual disability, myoclonus, and movement disorders [74,75].
SCN8A encephalopathy patients usually show severe developmental delay and intellectual disability, pharmacoresistant epilepsy with seizures starting before 18 months of age, in addition to pyramidal and extrapyramidal signs [76,77]. Epilepsy in SCN8A encephalopathy patients comprises multiple seizure types, including focal, generalized, and epileptic spasms [78]. Sudden unexpected death in epilepsy is reported in approximately 10% of cases [77].
As SCN8A epileptic encephalopathy is mostly due to gain-of-function mutations of SCN8A, the most effective AEDs reported so far are sodium channel blockers including phenytoin, carbamazepine, and oxcarbazepine, usually at supratherapeutic doses [77]. Also, at least one study reported improvements in seizure clusters and non-convulsive status epilepticus when patients were treated with benzodiazepines [77]. Of note, levetiracetam was reported to be ineffective and may even worsen seizure activity [77].
The syntaxin-binding protein 1 (STXBP1) gene encodes the STXBP1 protein, which is an essential protein for presynaptic vesicle docking and fusion through interaction with the soluble N-ethylmaleimide sensitive factor attachment protein receptor (SNARE) complex proteins, and has an essential function in neurotransmitter release [79,80]. STXBP1 encephalopathy is caused by both truncating and missense mutations in STXBP1, and the current leading hypothesis is that STXBP1 loss-of-function is the common pathomechanism, although some in vitro studies have reported possible dominant-negative effects or gain in pathological function [80-83].
STXBP1 encephalopathy patients show developmental delay/intellectual disability, epilepsy, autism spectrum disorders, and involuntary movements such as spasms and jerks [84,85]. Epilepsy in STXBP1 encephalopathy patients is characterized by early onset of tonic spasms, often immediately after birth, and drug-resistant seizures with multifocal epileptic activity on EEG [86].
Some case reports are currently available, and some beneficial AEDs include vigabatrin, valproic acid, and levetiracetam [81,82,87-91]. A beneficial effect of levetiracetam is noteworthy because it has been proposed to modulate synaptic vesicle release via binding with synaptic vesicle glycoprotein 2a [80,92]. However, there is insufficient data for this generalization. There is currently no drug on the market that directly targets STXBP1.
The synaptic RAS-GTPase-activating protein 1 (SYNGAP1) gene encodes SYNGAP1 protein, which is an important mediator in the N-methyl-D-aspartate (NMDA) receptor-activated RAS-signaling cascade that regulates postsynaptic density and formation, development, and maturation of dendritic spines [93,94]. In a mouse model, heterozygous mutations in Syngap1 led to protein truncations and abnormal dendritic spines during development [95], decreasing the ability of Syngap1 to bind downstream molecules in the signaling pathway, and thus failing to inhibit Ras activity. Increased Ras activation led to the activation of molecules that regulate the actin cytoskeleton, thus shifting the equilibrium towards the more stable actin form [93,96]. This, combined with an increase in insertion of AMPA receptors into the postsynaptic membrane during development, caused the dendritic spines to mature into mushroom-shaped spines earlier than normal, leading to elevated excitatory synaptic transmission and excitatory/inhibitory imbalance. The occurrence of these events during a critical period of development made the neurons more prone to seizures [93,97].
SYNGAP1 mutations cause developmental epileptic encephalopathy characterized by generalized epilepsy with eyelid myoclonia with absences and myoclonic-atonic seizures that are often pharmacoresistant [98]. Seizures triggered by eating is also a characteristic feature [98]. SYNGAP1 encephalopathy is associated with a spectrum of mild to severe intellectual disability, with a large proportion of patients with severe ID and other comorbidities such as behavioral problems, autism spectrum disorder, hypotonia, eating problems, and sleeping problems [98].
In a retrospective study with 57 patients, valproate (n=45) and lamotrigine (n=22) were the most commonly prescribed AEDs, with long-term treatment with lamotrigine in 77% and valproate in 64% of patients, respectively, suggesting effectiveness [98]. This is a reasonable result considering that generalized seizures occur in SYNGAP1-associated epilepsy patients and because there is currently no targeted therapy specifically for SYNGAP1.
Here, we have reviewed several developmental and epileptic encephalopathies caused by frequently identified genes. Already too well known SCN1A was not addressed. Dietary therapy and modulatory therapy such as vagus nerve stimulation were also not covered in this review. Therapies directly targeting at causative mutations, such as retigabine in KCNQ2 encephalopathy and quinidine in KCNT1 encephalopathy were discussed. However, despite promising results in vitro, the results were disappointing when administered to patients. Considering the pathogenic mechanisms of causative mutations, sodium channel blockers are recommended for patients with KCNQ2 mutations, infantile epileptic encephalopathy patients with SCN2A mutations, and patients with SCN8A mutations. Levetiracetam can be considered in patients with STXBP1 mutations (Table 1).
With a rapidly increasing number of genetically diagnosed patients, development of gene-specific therapies is in great demand. Studies to identify personalized therapeutic strategies based on functional studies of individuals will increasingly be required to meet this demand.
Table┬Ā1.
Summary of clinical presentations and recommended therapies by causative genes
| Gene | Clinical presentations | Medications |
|---|---|---|
| ALDH7A1 | Neonatal onset drug resistant seizures which respond dramatically to pyridoxine | Pyridoxine, with arginine supplementation and/or lysine restricted diet [9]. |
| Developmental delay/intellectual disability irrespective of pyridoxine supplementation | ||
| CDKL5 | Early infantile epilepsy (<2 months) with very frequent brief drug responsive generalized tonic seizures, followed by infantile spasms with hypsarrhythmia (around 11 months) that are drug resistant, and then drug resistant late multifocal and myoclonic epilepsy (in childhood) | No available medication reported to show significant beneficial effects. |
| KCNQ2 | Benign familial seizures of early infancy with seizures as early as within first days of life showing excellent prognosis regarding both seizure remission and neurodevelopment | Sodium channel blockers are recommended as first line treatment [31,34]. |
| Early infantile epileptic encephalopathy with drug resistant seizures as early as within first days of life, intellectual disability, and encephalopathic EEG | Levetiracetam or valproate can be used if patients do not respond to sodium channel blockers [31]. | |
| Retigabine is inferior to above medications [31]. | ||
| KCNT1 | EIMFS | Quinidine was not effective in ADNFLE patients [49]. |
| ADNFLE | Quinidine showed limited efficacy in EIMFS patients (responder rate=32%, n=25) [39,48,50-60]. | |
| SCN2A | Infantile epileptic encephalopathy with drug-resistant infantile-onset seizures (<12 months) and developmental delay | Sodium channel blockers are recommended for infantile epileptic encephalopathy and benign (familial) infantile seizure patients [62,63]. |
| Benign (familial) infantile seizures with infantile-onset seizures (<12 months) that resolve by 2 years of age without neurodevelopmental sequelae | Drugs other than sodium channel blockersŌĆölevetiracetam, benzodiazepines, and valproateŌĆöare recommended for autistic spectrum disorder/intellectual disability patients with childhood-onset seizures [61]. | |
| Autistic spectrum disorder/intellectual disability with global developmental delay, which 1/3 of patients develop childhood-onset seizures (>12 months) | ||
| SCN8A | Drug resistant epilepsy with seizure onset age of <18 months and multiple seizure types including focal seizures, generalized seizures, and epileptic spasms, developmental delay/intellectual disability, and pyramidal and extrapyramidal symptoms | Sodium channel blockers are recommended, but supratherapeutic doses can be needed [77]. |
| Levetiracetam can worsen the seizures [77]. | ||
| STXBP1 | Epilepsy with early onset tonic spasms (often immediately after birth) that are drug resistant, multifocal epileptic activity on EEG, developmental delay/intellectual disability, and involuntary movements such as spasms and jerks | Levetiracetam, vigabatrin, and valproic acid have been reported to be beneficial in small number of case reports [81,82,87-91]. |
| SYNGAP1 | Generalized epilepsy with eyelid myoclonia with absences and myoclonic-atonic seizures that are drug resistant, seizures triggered by eating, and developmental delay/intellectual disability | Valproate and lamotrigine are most frequently used [98]. |
ALDH7A1, aldehyde dehydrogenase 7 family, member A1; CDKL5, cyclin-dependent kinase-like 5; KCNQ2, potassium channel, voltage-gated, KQT-like subfamily member 2; EEG, electroencephalogram; KCNT1, potassium channel subfamily T member 1; EIMFS, epilepsy of infancy with migrating focal seizures; ADNFLE, autosomal dominant nocturnal frontal lobe epilepsy; SCN2A, sodium channel, voltage-gated, type II, alpha subunit; SCN8A, sodium channel, voltage-gated, type VIII alpha subunit; STXBP1, syntaxin-binding protein 1; SYNGAP1, synaptic RAS-GTPase-activating protein 1.
Fig.┬Ā1.
Number of publications on PubMed to search terms epilepsy, and personalized or precision medicine.
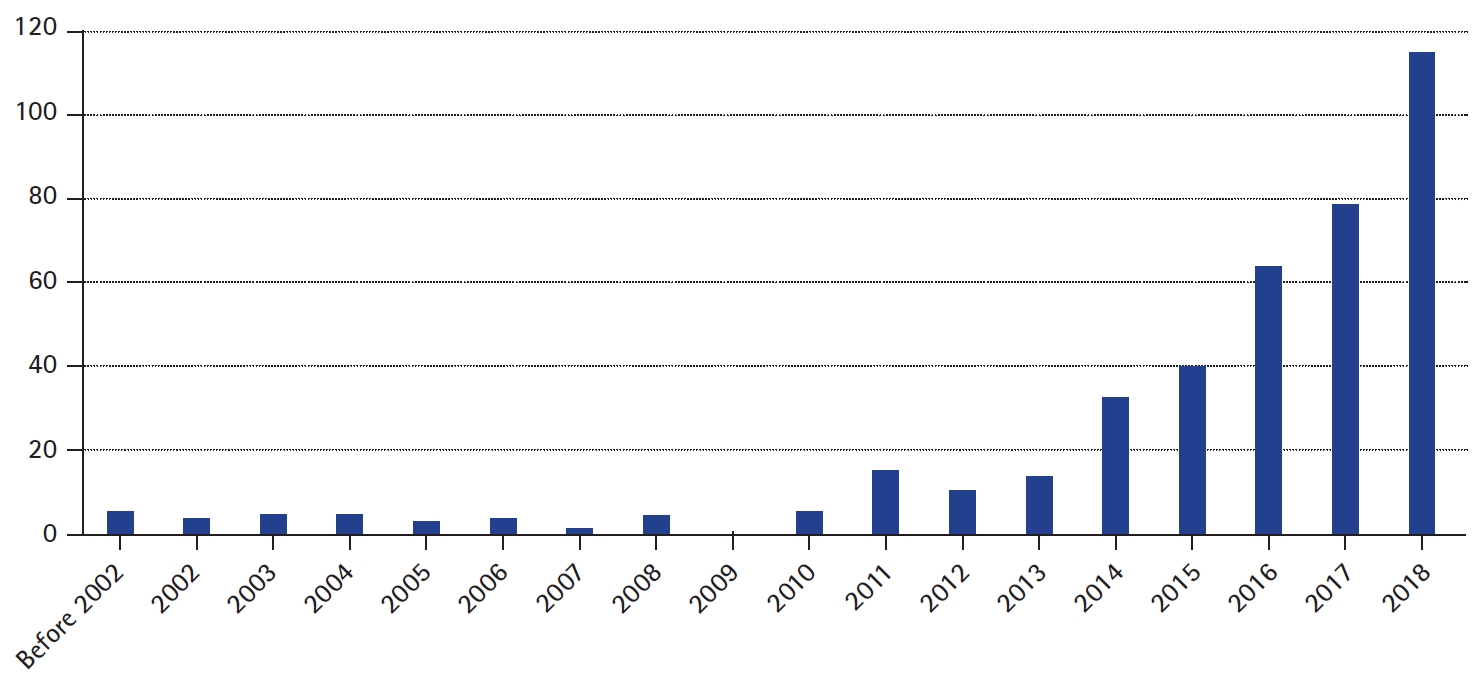
Fig.┬Ā2.
Model of sodium channel, voltage-gated, type II, alpha subunit (SCN2A) encephalopathy pathophysiology. Adapted from Sanders et al. [61], with permission from Elsevier.

References
1. Moller RS, Dahl HA, Helbig I. The contribution of next generation sequencing to epilepsy genetics. Expert Rev Mol Diagn 2015;15:1531-8.


2. Strzelczyk A, Klein KM, von Podewils F. Editorial: burden of illness in people with epilepsy: from population-based studies to precision medicine. Front Neurol 2019;9:1164.



3. Moller RS, Hammer TB, Rubboli G, Lemke JR, Johannesen KM. From next-generation sequencing to targeted treatment of non-acquired epilepsies. Expert Rev Mol Diagn 2019;19:217-28.

4. Mills PB, Struys E, Jakobs C, Plecko B, Baxter P, Baumgartner M, et al. Mutations in antiquitin in individuals with pyridoxine-dependent seizures. Nat Med 2006;12:307-9.



5. Coughlin CR 2nd, Swanson MA, Spector E, Meeks NJL, Kronquist KE, Aslamy M, et al. The genotypic spectrum of ALDH7A1 mutations resulting in pyridoxine dependent epilepsy: a common epileptic encephalopathy. J Inherit Metab Dis 2019;42:353-61.



6. Basura GJ, Hagland SP, Wiltse AM, Gospe SM Jr. Clinical features and the management of pyridoxine-dependent and pyridoxine-responsive seizures: review of 63 North American cases submitted to a patient registry. Eur J Pediatr 2009;168:697-704.


7. van Karnebeek CD, Hartmann H, Jaggumantri S, Bok LA, Cheng B, Connolly M, et al. Lysine restricted diet for pyridoxine-dependent epilepsy: first evidence and future trials. Mol Genet Metab 2012;107:335-44.


8. Mercimek-Mahmutoglu S, Cordeiro D, Cruz V, Hyland K, Struys EA, Kyriakopoulou L, et al. Novel therapy for pyridoxine dependent epilepsy due to ALDH7A1 genetic defect: L-arginine supplementation alternative to lysine-restricted diet. Eur J Paediatr Neurol 2014;18:741-6.


9. Coughlin CR 2nd, van Karnebeek CD, Al-Hertani W, Shuen AY, Jaggumantri S, Jack RM, et al. Triple therapy with pyridoxine, arginine supplementation and dietary lysine restriction in pyridoxine-dependent epilepsy: neurodevelopmental outcome. Mol Genet Metab 2015;116:35-43.


10. Mari F, Azimonti S, Bertani I, Bolognese F, Colombo E, Caselli R, et al. CDKL5 belongs to the same molecular pathway of MeCP2 and it is responsible for the early-onset seizure variant of Rett syndrome. Hum Mol Genet 2005;14:1935-46.



11. Ricciardi S, Ungaro F, Hambrock M, Rademacher N, Stefanelli G, Brambilla D, et al. CDKL5 ensures excitatory synapse stability by reinforcing NGL-1-PSD95 interaction in the postsynaptic compartment and is impaired in patient iPSC-derived neurons. Nat Cell Biol 2012;14:911-23.




12. Rusconi L, Salvatoni L, Giudici L, Bertani I, Kilstrup-Nielsen C, Broccoli V, et al. CDKL5 expression is modulated during neuronal development and its subcellular distribution is tightly regulated by the C-terminal tail. J Biol Chem 2008;283:30101-11.



13. Wang IT, Allen M, Goffin D, Zhu X, Fairless AH, Brodkin ES, et al. Loss of CDKL5 disrupts kinome profile and event-related potentials leading to autistic-like phenotypes in mice. Proc Natl Acad Sci U S A 2012;109:21516-21.



14. Okuda K, Kobayashi S, Fukaya M, Watanabe A, Murakami T, Hagiwara M, et al. CDKL5 controls postsynaptic localization of GluN2B-containing NMDA receptors in the hippocampus and regulates seizure susceptibility. Neurobiol Dis 2017;106:158-70.


15. Amendola E, Zhan Y, Mattucci C, Castroflorio E, Calcagno E, Fuchs C, et al. Mapping pathological phenotypes in a mouse model of CDKL5 disorder. PLoS One 2014;9:e91613.



16. Fuchs C, Trazzi S, Torricella R, Viggiano R, De Franceschi M, Amendola E, et al. Loss of CDKL5 impairs survival and dendritic growth of newborn neurons by altering AKT/GSK-3╬▓ signaling. Neurobiol Dis 2014;70:53-68.



17. Fuchs C, Rimondini R, Viggiano R, Trazzi S, De Franceschi M, Bartesaghi R, et al. Inhibition of GSK3╬▓ rescues hippocampal development and learning in a mouse model of CDKL5 disorder. Neurobiol Dis 2015;82:298-310.


18. Della Sala G, Putignano E, Chelini G, Melani R, Calcagno E, Michele Ratto G, et al. Dendritic spine instability in a mouse model of CDKL5 disorder is rescued by insulin-like growth factor 1. Biol Psychiatry 2016;80:302-11.


19. Pizzo R, Gurgone A, Castroflorio E, Amendola E, Gross C, Sassoe-Pognetto M, et al. Lack of Cdkl5 disrupts the organization of excitatory and inhibitory synapses and parvalbumin interneurons in the primary visual cortex. Front Cell Neurosci 2016;10:261.



20. Bahi-Buisson N, Kaminska A, Boddaert N, Rio M, Afenjar A, Gerard M, et al. The three stages of epilepsy in patients with CDKL5 mutations. Epilepsia 2008;49:1027-37.


21. Tao J, Van Esch H, Hagedorn-Greiwe M, Hoffmann K, Moser B, Raynaud M, et al. Mutations in the X-linked cyclin-dependent kinase-like 5 (CDKL5/STK9) gene are associated with severe neurodevelopmental retardation. Am J Hum Genet 2004;75:1149-54.



22. Devinsky O, Verducci C, Thiele EA, Laux LC, Patel AD, Filloux F, et al. Open-label use of highly purified CBD (Epidiolex®) in patients with CDKL5 deficiency disorder and Aicardi, Dup15q, and Doose syndromes. Epilepsy Behav 2018;86:131-7.


23. Tramarin M, Rusconi L, Pizzamiglio L, Barbiero I, Peroni D, Scaramuzza L, et al. The antidepressant tianeptine reverts synaptic AMPA receptor defects caused by deficiency of CDKL5. Hum Mol Genet 2018;27:2052-63.



24. Fuchs C, Fustini N, Trazzi S, Gennaccaro L, Rimondini R, Ciani E. Treatment with the GSK3-beta inhibitor Tideglusib improves hippocampal development and memory performance in juvenile, but not adult, Cdkl5 knockout mice. Eur J Neurosci 2018;47:1054-66.


25. Trazzi S, De Franceschi M, Fuchs C, Bastianini S, Viggiano R, Lupori L, et al. CDKL5 protein substitution therapy rescues neurological phenotypes of a mouse model of CDKL5 disorder. Hum Mol Genet 2018;27:1572-92.



26. Wang HS, Pan Z, Shi W, Brown BS, Wymore RS, Cohen IS, et al. KCNQ2 and KCNQ3 potassium channel subunits: molecular correlates of the M-channel. Science 1998;282:1890-3.


27. Jentsch TJ. Neuronal KCNQ potassium channels: physiology and role in disease. Nat Rev Neurosci 2000;1:21-30.



28. Miceli F, Soldovieri MV, Ambrosino P, De Maria M, Migliore M, Migliore R, et al. Early-onset epileptic encephalopathy caused by gain-of-function mutations in the voltage sensor of Kv7.2 and Kv7.3 potassium channel subunits. J Neurosci 2015;35:3782-93.



29. Millichap JJ, Miceli F, De Maria M, Keator C, Joshi N, Tran B, et al. Infantile spasms and encephalopathy without preceding neonatal seizures caused by KCNQ2 R198Q, a gain-of-function variant. Epilepsia 2017;58:e10-5.


30. Mulkey SB, Ben-Zeev B, Nicolai J, Carroll JL, Gronborg S, Jiang YH, et al. Neonatal nonepileptic myoclonus is a prominent clinical feature of KCNQ2 gain-of-function variants R201C and R201H. Epilepsia 2017;58:436-45.



31. Kuersten M, Tacke M, Gerstl L, Hoelz H, Stulpnagel CV, Borggraefe I. Antiepileptic therapy approaches in KCNQ2 related epilepsy: a systematic review. Eur J Med Genet 2019 Feb 14 [Epub]. https://doi.org/10.1016/j.ejmg.2019.02.001

32. Nguyen HM, Miyazaki H, Hoshi N, Smith BJ, Nukina N, Goldin AL, et al. Modulation of voltage-gated K+ channels by the sodium channel ╬▓1 subunit. Proc Natl Acad Sci U S A 2012;109:18577-82.



33. Pan Z, Kao T, Horvath Z, Lemos J, Sul JY, Cranstoun SD, et al. A common ankyrin-G-based mechanism retains KCNQ and NaV channels at electrically active domains of the axon. J Neurosci 2006;26:2599-613.



34. Pisano T, Numis AL, Heavin SB, Weckhuysen S, Angriman M, Suls A, et al. Early and effective treatment of KCNQ2 encephalopathy. Epilepsia 2015;56:685-91.


35. Barrese V, Miceli F, Soldovieri MV, Ambrosino P, Iannotti FA, Cilio MR, et al. Neuronal potassium channel openers in the management of epilepsy: role and potential of retigabine. Clin Pharmacol 2010;2:225-36.



36. Clark S, Antell A, Kaufman K. New antiepileptic medication linked to blue discoloration of the skin and eyes. Ther Adv Drug Saf 2015;6:15-9.



37. Ihara Y, Tomonoh Y, Deshimaru M, Zhang B, Uchida T, Ishii A, et al. Retigabine, a Kv7.2/Kv7.3-channel opener, attenuates drug-induced seizures in knock-in mice harboring Kcnq2 mutations. PLoS One 2016;11:e0150095.



38. Kaczmarek LK. Slack, slick and sodium-activated potassium channels. ISRN Neurosci 2013;2013:354262.




39. McTague A, Nair U, Malhotra S, Meyer E, Trump N, Gazina EV, et al. Clinical and molecular characterization of KCNT1-related severe early-onset epilepsy. Neurology 2018;90:e55-66.



40. Lim CX, Ricos MG, Dibbens LM, Heron SE. KCNT1 mutations in seizure disorders: the phenotypic spectrum and functional effects. J Med Genet 2016;53:217-25.


41. Barcia G, Fleming MR, Deligniere A, Gazula VR, Brown MR, Langouet M, et al. De novo gain-of-function KCNT1 channel mutations cause malignant migrating partial seizures of infancy. Nat Genet 2012;44:1255-9.




42. Milligan CJ, Li M, Gazina EV, Heron SE, Nair U, Trager C, et al. KCNT1 gain of function in 2 epilepsy phenotypes is reversed by quinidine. Ann Neurol 2014;75:581-90.



43. Ohba C, Kato M, Takahashi N, Osaka H, Shiihara T, Tohyama J, et al. De novo KCNT1 mutations in early-onset epileptic encephalopathy. Epilepsia 2015;56:e121-8.


44. Heron SE, Smith KR, Bahlo M, Nobili L, Kahana E, Licchetta L, et al. Missense mutations in the sodium-gated potassium channel gene KCNT1 cause severe autosomal dominant nocturnal frontal lobe epilepsy. Nat Genet 2012;44:1188-90.



45. Bhattacharjee A, Joiner WJ, Wu M, Yang Y, Sigworth FJ, Kaczmarek LK. Slick (Slo2.1), a rapidly-gating sodium-activated potassium channel inhibited by ATP. J Neurosci 2003;23:11681-91.



46. Yang B, Gribkoff VK, Pan J, Damagnez V, Dworetzky SI, Boissard CG, et al. Pharmacological activation and inhibition of Slack (Slo2.2) channels. Neuropharmacology 2006;51:896-906.


47. Kim GE, Kronengold J, Barcia G, Quraishi IH, Martin HC, Blair E, et al. Human slack potassium channel mutations increase positive cooperativity between individual channels. Cell Rep 2014;9:1661-72.



48. Mikati MA, Jiang YH, Carboni M, Shashi V, Petrovski S, Spillmann R, et al. Quinidine in the treatment of KCNT1-positive epilepsies. Ann Neurol 2015;78:995-9.



49. Mullen SA, Carney PW, Roten A, Ching M, Lightfoot PA, Churilov L, et al. Precision therapy for epilepsy due to KCNT1 mutations: a randomized trial of oral quinidine. Neurology 2018;90:e67-72.


50. Yoshitomi S, Takahashi Y, Yamaguchi T, Oboshi T, Horino A, Ikeda H, et al. Quinidine therapy and therapeutic drug monitoring in four patients with KCNT1 mutations. Epileptic Disord 2019;21:48-54.

51. Chen Y, Bao XH, Zhang QP, Wang JP, Wen YX, Yu SJ, et al. Genetic and clinical analysis of children with early-onset epilepsy encephalopathy caused by KCNT1 gene mutation. Zhonghua Er Ke Za Zhi 2018;56:824-8.


52. Numis AL, Nair U, Datta AN, Sands TT, Oldham MS, Patel A, et al. Lack of response to quinidine in KCNT1-related neonatal epilepsy. Epilepsia 2018;59:1889-98.


53. Dilena R, DiFrancesco JC, Soldovieri MV, Giacobbe A, Ambrosino P, Mosca I, et al. Early treatment with quinidine in 2 patients with epilepsy of infancy with migrating focal seizures (EIMFS) due to gain-of-function KCNT1 mutations: functional studies, clinical responses, and critical issues for personalized therapy. Neurotherapeutics 2018;15:1112-26.




54. Ambrosino P, Soldovieri MV, Bast T, Turnpenny PD, Uhrig S, Biskup S, et al. De novo gain-of-function variants in KCNT2 as a novel cause of developmental and epileptic encephalopathy. Ann Neurol 2018;83:1198-204.


55. Ko A, Youn SE, Kim SH, Lee JS, Kim S, Choi JR, et al. Targeted gene panel and genotype-phenotype correlation in children with developmental and epileptic encephalopathy. Epilepsy Res 2018;141:48-55.


56. Abdelnour E, Gallentine W, McDonald M, Sachdev M, Jiang YH, Mikati MA. Does age affect response to quinidine in patients with KCNT1 mutations? Report of three new cases and review of the literature. Seizure 2018;55:1-3.


57. Madaan P, Jauhari P, Gupta A, Chakrabarty B, Gulati S. A quinidine non responsive novel KCNT1 mutation in an Indian infant with epilepsy of infancy with migrating focal seizures. Brain Dev 2018;40:229-32.


58. Fukuoka M, Kuki I, Kawawaki H, Okazaki S, Kim K, Hattori Y, et al. Quinidine therapy for West syndrome with KCNTI mutation: a case report. Brain Dev 2017;39:80-3.


59. Chong PF, Nakamura R, Saitsu H, Matsumoto N, Kira R. Ineffective quinidine therapy in early onset epileptic encephalopathy with KCNT1 mutation. Ann Neurol 2016;79:502-3.


60. Bearden D, Strong A, Ehnot J, DiGiovine M, Dlugos D, Goldberg EM. Targeted treatment of migrating partial seizures of infancy with quinidine. Ann Neurol 2014;76:457-61.


61. Sanders SJ, Campbell AJ, Cottrell JR, Moller RS, Wagner FF, Auldridge AL, et al. Progress in understanding and treating SCN2A-mediated disorders. Trends Neurosci 2018;41:442-56.



62. Ben-Shalom R, Keeshen CM, Berrios KN, An JY, Sanders SJ, Bender KJ. Opposing effects on Na(V)1.2 function underlie differences between SCN2A variants observed in individuals with autism spectrum disorder or infantile seizures. Biol Psychiatry 2017;82:224-32.



63. Wolff M, Johannesen KM, Hedrich UBS, Masnada S, Rubboli G, Gardella E, et al. Genetic and phenotypic heterogeneity suggest therapeutic implications in SCN2A-related disorders. Brain 2017;140:1316-36.



65. Lorincz A, Nusser Z. Cell-type-dependent molecular composition of the axon initial segment. J Neurosci 2008;28:14329-40.



66. Caldwell JH, Schaller KL, Lasher RS, Peles E, Levinson SR. Sodium channel Na(v)1.6 is localized at nodes of ranvier, dendrites, and synapses. Proc Natl Acad Sci U S A 2000;97:5616-20.



67. Bunton-Stasyshyn RKA, Wagnon JL, Wengert ER, Barker BS, Faulkner A, Wagley PK, et al. Prominent role of forebrain excitatory neurons in SCN8A encephalopathy. Brain 2019;142:362-75.




68. Hu W, Tian C, Li T, Yang M, Hou H, Shu Y. Distinct contributions of Na(v)1.6 and Na(v)1.2 in action potential initiation and backpropagation. Nat Neurosci 2009;12:996-1002.



69. Veeramah KR, O'Brien JE, Meisler MH, Cheng X, Dib-Hajj SD, Waxman SG, et al. De novo pathogenic SCN8A mutation identified by whole-genome sequencing of a family quartet affected by infantile epileptic encephalopathy and SUDEP. Am J Hum Genet 2012;90:502-10.



70. de Kovel CG, Meisler MH, Brilstra EH, van Berkestijn FM, van 't Slot R, van Lieshout S, et al. Characterization of a de novo SCN8A mutation in a patient with epileptic encephalopathy. Epilepsy Res 2014;108:1511-8.



71. Estacion M, O'Brien JE, Conravey A, Hammer MF, Waxman SG, Dib-Hajj SD, et al. A novel de novo mutation of SCN8A (Nav1.6) with enhanced channel activation in a child with epileptic encephalopathy. Neurobiol Dis 2014;69:117-23.



72. Baker EM, Thompson CH, Hawkins NA, Wagnon JL, Wengert ER, Patel MK, et al. The novel sodium channel modulator GS-458967 (GS967) is an effective treatment in a mouse model of SCN8A encephalopathy. Epilepsia 2018;59:1166-76.



73. Patel RR, Barbosa C, Brustovetsky T, Brustovetsky N, Cummins TR. Aberrant epilepsy-associated mutant Nav1.6 sodium channel activity can be targeted with cannabidiol. Brain 2016;139:2164-81.




74. Wagnon JL, Barker BS, Ottolini M, Park Y, Volkheimer A, Valdez P, et al. Loss-of-function variants of SCN8A in intellectual disability without seizures. Neurol Genet 2017;3:e170.



75. Wagnon JL, Mencacci NE, Barker BS, Wengert ER, Bhatia KP, Balint B, et al. Partial loss-of-function of sodium channel SCN8A in familial isolated myoclonus. Hum Mutat 2018;39:965-9.



76. Ohba C, Kato M, Takahashi S, Lerman-Sagie T, Lev D, Terashima H, et al. Early onset epileptic encephalopathy caused by de novo SCN8A mutations. Epilepsia 2014;55:994-1000.


77. Gardella E, Marini C, Trivisano M, Fitzgerald MP, Alber M, Howell KB, et al. The phenotype of SCN8A developmental and epileptic encephalopathy. Neurology 2018;91:e1112-24.


78. O'Brien JE, Meisler MH. Sodium channel SCN8A (Nav1.6): properties and de novo mutations in epileptic encephalopathy and intellectual disability. Front Genet 2013;4:213.



79. Pevsner J, Hsu SC, Scheller RH. n-Sec1: a neural-specific syntaxin-binding protein. Proc Natl Acad Sci U S A 1994;91:1445-9.



80. Stamberger H, Weckhuysen S, De Jonghe P. STXBP1 as a therapeutic target for epileptic encephalopathy. Expert Opin Ther Targets 2017;21:1027-36.


81. Stamberger H, Nikanorova M, Willemsen MH, Accorsi P, Angriman M, Baier H, et al. STXBP1 encephalopathy: a neurodevelopmental disorder including epilepsy. Neurology 2016;86:954-62.


82. Mignot C, Moutard ML, Trouillard O, Gourfinkel-An I, Jacquette A, Arveiler B, et al. STXBP1-related encephalopathy presenting as infantile spasms and generalized tremor in three patients. Epilepsia 2011;52:1820-7.


83. Chai YJ, Sierecki E, Tomatis VM, Gormal RS, Giles N, Morrow IC, et al. Munc18-1 is a molecular chaperone for ╬▒-synuclein, controlling its self-replicating aggregation. J Cell Biol 2016;214:705-18.



84. Hamdan FF, Piton A, Gauthier J, Lortie A, Dubeau F, Dobrzeniecka S, et al. De novo STXBP1 mutations in mental retardation and nonsyndromic epilepsy. Ann Neurol 2009;65:748-53.


85. De Rubeis S, He X, Goldberg AP, Poultney CS, Samocha K, Cicek AE, et al. Synaptic, transcriptional and chromatin genes disrupted in autism. Nature 2014;515:209-15.




86. Saitsu H, Kato M, Okada I, Orii KE, Higuchi T, Hoshino H, et al. STXBP1 mutations in early infantile epileptic encephalopathy with suppression-burst pattern. Epilepsia 2010;51:2397-405.


87. Deprez L, Weckhuysen S, Holmgren P, Suls A, Van Dyck T, Goossens D, et al. Clinical spectrum of early-onset epileptic encephalopathies associated with STXBP1 mutations. Neurology 2010;75:1159-65.


88. Milh M, Villeneuve N, Chouchane M, Kaminska A, Laroche C, Barthez MA, et al. Epileptic and nonepileptic features in patients with early onset epileptic encephalopathy and STXBP1 mutations. Epilepsia 2011;52:1828-34.


89. Mastrangelo M, Peron A, Spaccini L, Novara F, Scelsa B, Introvini P, et al. Neonatal suppression-burst without epileptic seizures: expanding the electroclinical phenotype of STXBP1-related, early-onset encephalopathy. Epileptic Disord 2013;15:55-61.


90. Romaniello R, Zucca C, Tenderini E, Arrigoni F, Ragona F, Zorzi G, et al. A novel mutation in STXBP1 gene in a child with epileptic encephalopathy and an atypical electroclinical pattern. J Child Neurol 2014;29:249-53.


91. Dilena R, Striano P, Traverso M, Viri M, Cristofori G, Tadini L, et al. Dramatic effect of levetiracetam in early-onset epileptic encephalopathy due to STXBP1 mutation. Brain Dev 2016;38:128-31.


92. Lynch BA, Lambeng N, Nocka K, Kensel-Hammes P, Bajjalieh SM, Matagne A, et al. The synaptic vesicle protein SV2A is the binding site for the antiepileptic drug levetiracetam. Proc Natl Acad Sci U S A 2004;101:9861-6.



95. Walkup WG 4th, Washburn L, Sweredoski MJ, Carlisle HJ, Graham RL, Hess S, et al. Phosphorylation of synaptic GTPase-activating protein (synGAP) by Ca2+/calmodulin-dependent protein kinase II (CaMKII) and cyclin-dependent kinase 5 (CDK5) alters the ratio of its GAP activity toward Ras and Rap GTPases. J Biol Chem 2015;290:4908-27.


96. Dillon C, Goda Y. The actin cytoskeleton: integrating form and function at the synapse. Annu Rev Neurosci 2005;28:25-55.


- TOOLS







