|
 |
- Search
| Ann Child Neurol > Volume 28(1); 2020 > Article |
|
Abstract
Benign convulsions with mild gastroenteritis (CwG) are a well-known type of benign seizures that occur in infants and children aged between 6 months and 3 years and are associated with acute (viral) gastroenteritis. The pathogens found in the stool of CwG patients are mostly rotavirus or norovirus, which can result in mild dehydration. Short-lasting seizures (≤ 5 minutes) occur in clusters within 24 hours, without provoking features such as fever, abnormal laboratory findings in the blood (e.g., hypoglycemia, hyponatremia, or hypocalcemia), or abnormal results in the cerebrospinal fluid (e.g., central nervous system infection). Electroencephalography in CwG patients shows normal or mildly abnormal findings, and brain imaging findings are normal. Affected children develop normally before and after the seizures. The occurrence of seizures does not require the repeated use of first- or second-line intravenous antiepileptic drugs in the acute stage or daily antiepileptic drug medications, since they usually do not recur and have a good prognosis. Patients with CwG rarely have a family history of epilepsy. Although the mean interval between enteric symptom onset and seizure onset in CwG is roughly 2 days, some patients can experience seizures before enteric symptoms, meaning that clinicians should exercise caution during early winter and spring, when the prevalence of CwG is especially high. Additionally, reports of CwG in Korea are roughly as common as in Japan and other East Asian countries, and pediatricians should therefore be familiar with its clinical characteristics and take care not to overprescribe antiepileptic drugs in patients with CwG.
Seizures in infancy and early childhood are frequently a major problem for the families of patients, especially if they are not simple febrile seizures with a good prognosis. If an infant younger than 6 months developed normally before the onset of the first seizure and experiences short-lasting afebrile seizures occurring once or twice a day, a clinician can first consider benign infantile seizures [1]. However, when the patient has symptoms of diarrhea and/or vomiting before or after seizures, this can indicate the presence of benign convulsions with mild gastroenteritis (CwG) [2-31]. Additionally, if the season is early winter or spring when rotavirus and norovirus are prevalent, CwG have to be considered in patients especially between 13 to 24 months of age with short-lasting seizures (less than 5 minutes) occurring in clusters [3-12,15,16,19,24].
CwG are benign convulsions that occur between the ages of 6 months and 3 years, with this age range first defined by Komori et al. [3] in 1995. However, the age range of patients with CwG has been estimated in the literature as being as broad as from 1 month to 6 years [2-20,22-26,30,31]. Hypoglycemia, an electrolyte imbalance such as hyponatremia or hypocalcemia, and central nervous system infection that can trigger seizures have to be excluded in this definition [2-31]. The acute gastroenteritis in these benign convulsions is mostly caused by rotavirus or norovirus [2-11,14-20,22-26]. Since CwG have been reported as frequently in Korea as in Japan, pediatricians need a good knowledge of their clinical characteristics and treatment [4-7,28,29].
The Japanese clinician Morooka [2] first reported patients with CwG in 1982. There were few reports of CwG in East Asian countries around Japan until the early 2000s [2,3,9-11], but it has been widely detected even in Western countries since the mid-2000s [12,17,19,20]. Following the first report by Morooka [2], Komori et al. [3] summarized the characteristics of CwG and tried to define this condition in 1995. Although there is still no categorized and constant definition, CwG can be easily recognized based on previously reported concepts: (1) benign seizures in children typically aged between 6 months and 3 years (or maximally between 1 month and 6 years); (2) normal development before and after seizures; (3) afebrile seizures with acute (viral) gastroenteritis that usually occur in winter and can cause mild dehydration, with the pathogens mostly being rotavirus or norovirus; (4) short-lasting recurrent seizures occurring within 24 hours (maximally within a few days); (5) neither laboratory abnormalities such as hypoglycemia and electrolyte imbalance nor abnormal findings in cerebrospinal fluid; (6) normal or mildly abnormal findings in electroencephalography (EEG); (7) normal findings in brain imaging; and (8) seizures having a good prognosis [2-31]. Relatively few patients with CwG have a family history of epilepsy [3,10,24-26,30].
The infectious pathogens of gastroenteritis found in patients with CwG are rotavirus, norovirus, adenovirus, astrovirus, and sapovirus [3-11,14-20,22-26]. Rotavirus is the most frequently reported, occurring in 40% to 50% patients with CwG before the rotavirus vaccine was introduced [3,5,10,15,17,23,24]. Rotavirus vaccines were introduced in South Korea in 2007, with the vaccination rate reaching 50% in 2009 [32,33]. These vaccines have decreased the rate of rotavirus positivity in patients with CwG, but the total incidence of CwG did not decrease since the incidence of norovirus infections has increased to become the leading pathogen in recent years [4-6]. Two Korean single-center studies performed after 2012 revealed 63.0% to 67.5% positivity for norovirus in patients with CwG [4,6]. The descriptions of the clinical characteristics have differed somewhat between rotaviral and noroviral CwG in a few studies; however, these had some limitations due to the small numbers of patients compared and the different prevalence periods of the two pathogens [4,5,15]. Afebrile seizures occur more frequently in patients with noroviral gastroenteritis than in those with rotaviral gastroenteritis, according to studies performed in Taiwan (29.7% vs. 5%) and Hong Kong (8.67% vs. 1.29%) [16,21].
The pathophysiologic mechanisms underlying seizures in patients with CwG are still unclear. The following two questions remain unanswered [7,8]: (1) are seizures in CwG caused by the direct invasion of viruses into the central nervous system or are they an indirect effect of viruses on the brain with circulating mediates such as cytokines, and (2) why are only infants and young children susceptible to CwG? Rotavirus mainly replicating in the gastrointestinal tracts has been detected in blood, which suggests possibility of the viral spread to the brain [34-39]. Rotavirus nonstructural protein 4 (NSP4) that is an important glycosylated protein for viral pathogenicity has been detected in rotavirus-infected neurons [36]. NSP4 has been mentioned as a major enterotoxin causing neurotoxicity by direct viral invasion or cytokine dysregulation [7,8,37]. Recently, Yeom et al. [37] detected that serum levels of anti-NSP4 immunoglobulin G (IgG) antibodies were lower in the seizure group with rotaviral gastroenteritis than those in the non-seizure group, which suggested the protective effect of anti-NSP4 IgG against seizures. The pathogenesis of norovirus and other viruses resulting in CwG has been still not-well understood [7,8,40]. Therefore, common hypothesis explaining pathogenic mechanism in CwG has not been proposed yet.
Rotavirus has been reported in more severe neurologic diseases than CwG: encephalopathy (e.g., mild encephalopathy with a reversible splenial lesion [MERS]), meningoencephalitis and cerebellitis [7,8,38,39]. Norovirus also has been detected in patients with encephalitis/encephalopathy [40]. A recent nationwide survey in Japan from 2011 to March 2016 showed how poor the outcome of norovirus-associated encephalitis/encephalopathy (NoVE) was in children [40]. NoVE in this survey included MERS, acute necrotizing encephalopathy, acute encephalopathy with biphasic seizures and late reduced diffusion, and hemorrhagic shock and encephalopathy [40].
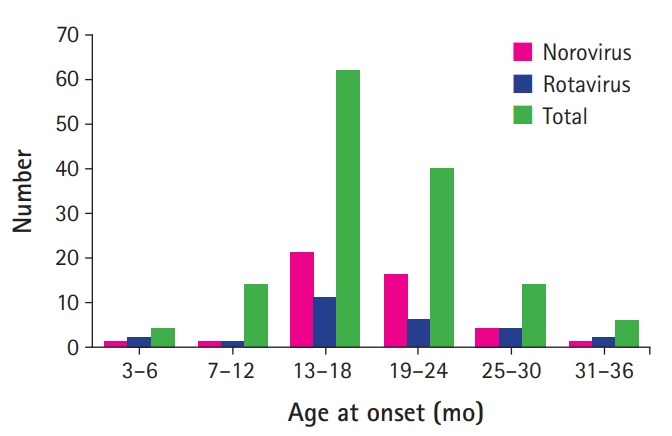
The incidence of CwG peaked at an age of 13 to 24 months regardless of the types of viruses involved (Fig. 1) [3-20,22-26]. The study of Kawano et al. [15] from 2007 found that nine patients with noroviral CwG were younger than 30 patients with rotaviral CwG. However, recent studies have found no age difference between noroviral CwG patients and rotaviral CwG patients (Table 1) [4,6]. A nonsignificant sex difference has been found in patients with CwG, with females constituting 55% to 60% of them [4,6,7,11,15]. A female predominance seemed more likely in patients with noroviral CwG (61.4% to 68.9%) than those with rotaviral CwG (50.0% to 53.5%) [4,6,15].
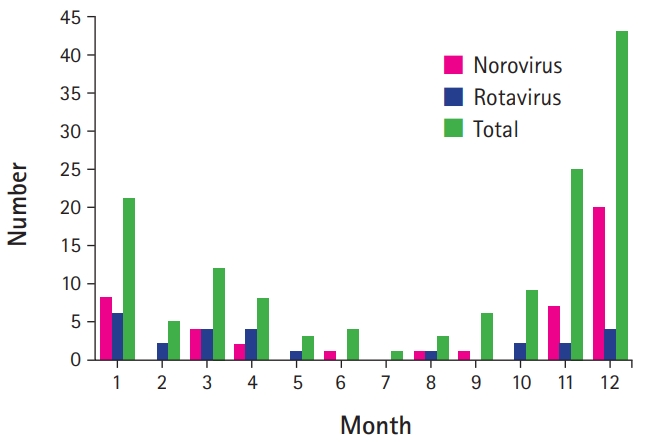
The most-common season for CwG is winter, independent of the virus type [3-12,15,16,18,19,24]. However, rotaviral CwG were most prevalent from January to May in East Asian countries (from winter to spring), while noroviral CwG were most prevalent during November and December (only in winter) (Fig. 2) [4,15,24].
CwG occur in distinctive clusters, which were noted in 57% to 75% of patients and reportedly involved a maximum of eight episodes in each cluster [3,5,7,10,15,17,24,30]. In the study of Kim et al. [4], multiple seizures occurred in 79.5% of the noroviral patients but 57.7% of the rotaviral patients (P=0.05) (Table 1). The number of seizures was 2.41±1.35 for the 140 CwG patients in this study: 2.73±1.37 in the noroviral group (44 patients) and 2.15±1.16 in the rotaviral group (26 patients; P=0.08) (Table 1) [4]. Another Korean study of noroviral CwG similarly found that the number of seizures was 2.79±2.82 [6].
One seizure episode lasts mostly for shorter than 5 minutes, although a few episodes last 15 minutes (Table 1) [4,10,15]. All seizure episodes from the first to the last mostly occur within 24 hours [4,10,15]: Kim et al.’s [4] recent study found that 62.1% of seizure episodes stopped within 6 hours, 87.1% within 12 hours, and 96.4% within 24 hours (Fig. 3). Although Kawano et al. [15] reported that all episodes of clustered seizures stopped earlier in rotaviral patients than in noroviral patients group (4.9±5.7 hours vs. 11.8±12.0 hours), this result was not replicated in Kim et al.’s [4] study.
The interval between enteric symptom onset and seizure onset in CwG was 2.11±1.14 days according to Kim et al.’s [4] study that included 140 CwG patients (Table 1). This interval was 2.00±1.06 days in 44 noroviral CwG patients but 2.58±1.21 days in 26 rotaviral CwG patients (P=0.04) (Table 1), although Kawano et al. [15] found no significant difference between the two groups. A recent study from 2016 similarly found that this interval in noroviral CwG was 1.82±0.78 days [6]. Some patients can experience seizures before enteric symptoms, although most CwG patients had seizures after enteric symptoms (Table 1) [3-5], and so clinicians should exercise caution during the prevalent season of CwG. Vomiting was more common in norovirus-associated CwG patients (97.7%) than in rotavirus-associated CwG patients (80.8%) in Kim et al.’s [4] previous study, although vomiting was usually less frequent in noroviral gastroenteritis than in rotaviral gastroenteritis (Table 1) [16]. This needs further investigation.
Seizure semiology of CwG has been differently described in the literature [3,4,6,17]. In two recent Korean studies, generalized onset seizures were seen in more than 90% of CwG patients irrespective of the virus type [4,6]. However, focal onset seizures were not less common than generalized onset ones in the studies of Komori et al. [3] (52.6%; 10 of 19 seizures in 10 CwG patients) and Caraballo et al. [17] (68.5%; 15 of 22 CwG patients). In fact, seizures in most ictal EEG recordings were focal onset ones [19,24].
The interictal EEG recordings in CwG are mostly normal, although they can show posterior slowing or focal sharp/spikes [3-8,10-12,15-20,24-26]. Kim et al.’s [4] previous study found that posterior slowing was more frequent in norovirus patients (34.9%) than in rotavirus patients (11.5%), while another Korean study of noroviral CwG observed focal or diffuse slowing in only 13.8% patients [4,6]. The findings of brain imaging were all normal in Kim et al.’s [4] previous study as well as in most other previous studies [4-8,15-20,24-26].
Most laboratory profiles in patients with CwG are within the normal ranges, but there are some reports of serum uric acid levels being high, including Kim et al.’s [4] recent report (9.53±0.48 mg/dL in 26 CwG patients) [4,27-29]. Yoo et al. [29] identified that serum uric acid levels in 154 patients with CwG were significantly higher (9.79±2.16 mg/dL) than in 3092 patients with acute gastroenteritis without seizures (6.04±2.36 mg/dL). Additionally, they showed that the serum levels of uric acid in CwG patients are not confounded by dehydration or recurrent seizures that have been considered as the underlying patho-mechanisms of high uric acid levels [29]. Yoo et al. [29] suggested that significantly-high serum uric acid levels can be a valuable diagnostic clues for CwG, although further basic studies to reveal its mechanism are in need. Comparisons of laboratory profiles between rotaviral and noroviral CwG patients in the literature have shown different results, which therefore need to be evaluated further with larger numbers of patients [4,15]. Although Kawano et al. [15] revealed no significant difference, Kim et al.’s [4] recent study showed a slight difference, with a higher platelet count (318,090±88,920/mm3 vs. 263,080±97,070/mm3, P=0.02), lower serum glucose level (76.86±14.96 mg/dL vs. 86.58±18.65 mg/dL, P=0.02), and higher serum calcium level (9.65±0.59 mg/dL vs. 9.41±0.37 mg/dL, P=0.04) in the patients with noroviral CwG than in those with rotaviral CwG.
Treating CwG in the acute stage when seizures develop in clusters does not require repeated injections of first- and second-line antiepileptic drugs, as seizures in CwG patients are characteristically short-lasting (≤5 minutes/episode) and all episodes of clustered seizures usually stop within 24 hours from seizure onset [10,14,15,23]. According to Japanese colleagues’ reports, benzodiazepines are not so effective, while carbamazepine (orally 5 mg/kg once daily for 1 to 3 days) and lidocaine have been effective in some patients with recurrent seizures [10,14,15,23]. Additionally, no daily antiepileptic drug medication is required since the seizures usually do not recur [10,14,15,23].
CwG are afebrile, nonprovoked, and archetypal seizures that occur during infancy and early childhood and have a good prognosis, and are associated with acute viral gastroenteritis and no abnormal findings in brain imaging. The most common pathogen was rotavirus before the rotavirus vaccine was introduced around 2010, and has been norovirus since then, with both of these viruses being most prevalent during winter. Short-lasting and clustered seizures occur within 24 hours, but they need neither repeated use of first- and second-line intravenous antiepileptic drugs in the acute stage nor daily antiepileptic drug medication. EEG in CwG patients shows normal or only mildly abnormal findings. The children develop normally before and after seizures and most of them have no specific family history of seizures. Although most seizures develop within 2 days after the onset of enteric symptoms, pediatricians should exercise caution during the prevalent season of CwG since seizures can also precede the enteric symptoms.
Notes
Author contributions
Conceptualization: YOK. Data curation: YOK. Formal analysis: YOK. Project administration: YOK. Visualization: YOK. Writing-original draft: YOK. Writing-review & editing: YOK.
Table 1.
| Characteristic | Norovirus | Rotavirus | Total | P valuea |
|---|---|---|---|---|
| Total number | 44 | 26 | 140 | |
| Age (mo) | 18.66±5.57 | 19.31±7.37 | 18.45±6.22 | 0.68 |
| Female sex | 27 (61.4) | 13 (50.0) | 77 (55.0) | 0.36 |
| Enteric symptoms | ||||
| Present at seizure onset | 44 (100.0) | 26 (100.0) | 140 (100.0) | |
| Diarrhea | 34 (77.3) | 23 (88.5) | 116 (82.9) | 0.25 |
| Vomiting | 43 (97.7) | 21 (80.8) | 120 (85.7) | 0.02c |
| Interval (day)b | 2.00±1.06 | 2.58±1.21 | 2.11±1.14 | 0.04c |
| Number of seizures | 2.73±1.37 | 2.15±1.16 | 2.41±1.35 | 0.08 |
| 1 | 9 (20.5) | 11 (42.3) | 44 (31.4) | 0.05 |
| 2 | 12 (27.3) | 4 (15.4) | 34 (24.3) | 0.26 |
| 3 | 11 (25.0) | 7 (26.9) | 39 (27.9) | 0.86 |
| 4 | 8 (18.2) | 4 (15.4) | 13 (9.3) | 0.77 |
| ≥5 | 4 (9.1) | 0 | 10 (7.1) | 0.12 |
| Seizure duration (min) | ||||
| ≤5 | 42 (95.5) | 24 (92.3) | 129 (92.1) | 0.58 |
| 6-15 | 2 (4.5) | 2 (7.7) | 9 (6.4) | 0.58 |
| >15 | 0 | 0 | 2 (1.4) |
References
1. Specchio N, Vigevano F. The spectrum of benign infantile seizures. Epilepsy Res 2006;70 Suppl 1:S156-67.


2. Morooka K. Convulsions and mild diarrhea. Shonika (Tokyo) 1982;23:131-7.
3. Komori H, Wada M, Eto M, Oki H, Aida K, Fujimoto T. Benign convulsions with mild gastroenteritis: a report of 10 recent cases detailing clinical varieties. Brain Dev 1995;17:334-7.


4. Kim BR, Choi GE, Kim YO, Kim MJ, Song ES, Woo YJ. Incidence and characteristics of norovirus-associated benign convulsions with mild gastroenteritis, in comparison with rotavirus ones. Brain Dev 2018;40:699-706.


5. Park SH, Kim YO, Kim HK, Kim HS, Kim BY, Cheon KR, et al. Incidence of benign convulsions with mild gastroenteritis after introduction of rotavirus vaccine. Brain Dev 2015;37:625-30.


6. Kim GH, Byeon JH, Lee DY, Jeong HJ, Eun BL. Norovirus in benign convulsions with mild gastroenteritis. Ital J Pediatr 2016;42:94.




8. Castellazzi L, Principi N, Agostoni C, Esposito S. Benign convulsions in children with mild gastroenteritis. Eur J Paediatr Neurol 2016;20:690-5.


9. Abe T, Kobayashi M, Araki K, Kodama H, Fujita Y, Shinozaki T, et al. Infantile convulsions with mild gastroenteritis. Brain Dev 2000;22:301-6.


10. Uemura N, Okumura A, Negoro T, Watanabe K. Clinical features of benign convulsions with mild gastroenteritis. Brain Dev 2002;24:745-9.


11. Hung JJ, Wen HY, Yen MH, Chen HW, Yan DC, Lin KL, et al. Rotavirus gastroenteritis associated with afebrile convulsion in children: clinical analysis of 40 cases. Chang Gung Med J 2003;26:654-9.

12. Narchi H. Benign afebrile cluster convulsions with gastroenteritis: an observational study. BMC Pediatr 2004;4:2.




13. Okumura A, Uemura N, Negoro T, Watanabe K. Efficacy of antiepileptic drugs in patients with benign convulsions with mild gastroenteritis. Brain Dev 2004;26:164-7.


14. Okumura A, Tanabe T, Kato T, Hayakawa F, Watanabe K. A pilot study on lidocaine tape therapy for convulsions with mild gastroenteritis. Brain Dev 2004;26:525-9.


15. Kawano G, Oshige K, Syutou S, Koteda Y, Yokoyama T, Kim BG, et al. Benign infantile convulsions associated with mild gastroenteritis: a retrospective study of 39 cases including virological tests and efficacy of anticonvulsants. Brain Dev 2007;29:617-22.


16. Chen SY, Tsai CN, Lai MW, Chen CY, Lin KL, Lin TY, et al. Norovirus infection as a cause of diarrhea-associated benign infantile seizures. Clin Infect Dis 2009;48:849-55.



17. Caraballo RH, Ganez L, Santos Cde L, Espeche A, Cersosimo R, Fejerman N. Benign infantile seizures with mild gastroenteritis: study of 22 patients. Seizure 2009;18:686-9.


18. Verrotti A, Tocco AM, Coppola GG, Altobelli E, Chiarelli F. Afebrile benign convulsions with mild gastroenteritis: a new entity? Acta Neurol Scand 2009;120:73-9.


19. Cusmai R, Jocic-Jakubi B, Cantonetti L, Japaridze N, Vigevano F. Convulsions associated with gastroenteritis in the spectrum of benign focal epilepsies in infancy: 30 cases including four cases with ictal EEG recording. Epileptic Disord 2010;12:255-61.



20. Lloyd MB, Lloyd JC, Gesteland PH, Bale JF Jr. Rotavirus gastroenteritis and seizures in young children. Pediatr Neurol 2010;42:404-8.


21. Chan CM, Chan CW, Ma CK, Chan HB. Norovirus as cause of benign convulsion associated with gastro-enteritis. J Paediatr Child Health 2011;47:373-7.


22. Dura-Trave T, Yoldi-Petri ME, Gallinas-Victoriano F, Molins-Castiella T. Infantile convulsions with mild gastroenteritis: a retrospective study of 25 patients. Eur J Neurol 2011;18:273-8.


23. Tanabe T, Okumura A, Komatsu M, Kubota T, Nakajima M, Shimakawa S. Clinical trial of minimal treatment for clustering seizures in cases of convulsions with mild gastroenteritis. Brain Dev 2011;33:120-4.


24. Verrotti A, Nanni G, Agostinelli S, Parisi P, Capovilla G, Beccaria F, et al. Benign convulsions associated with mild gastroenteritis: a multicenter clinical study. Epilepsy Res 2011;93:107-14.


25. Li T, Hong S, Peng X, Cheng M, Jiang L. Benign infantile convulsions associated with mild gastroenteritis: an electroclinical study of 34 patients. Seizure 2014;23:16-9.


26. Ueda H, Tajiri H, Kimura S, Etani Y, Hosoi G, Maruyama T, et al. Clinical characteristics of seizures associated with viral gastroenteritis in children. Epilepsy Res 2015;109:146-54.


27. Tsujita Y, Matsumoto H, Nakamura Y, Nonoyama S. Analysis of the blood and serum biochemistry findings in patients demonstrating convulsion with mild gastroenteritis. No To Hattatsu 2011;43:282-4.

28. Chae SH, Rhee M, Kim YC, Kim SS. The relationship between serum uric acid level and benign convulsions with mild gastroenteritis. J Korean Child Neurol Soc 2014;22:191-4.

29. Yoo IH, Kim W, Cho J, Kim H, Lim BC, Hwang H, et al. Elevated serum uric acid in benign convulsions with mild gastroenteritis in children. J Clin Neurol 2019;15:496-501.



30. Verrotti A, Moavero R, Vigevano F, Cantonetti L, Guerra A, Spezia E, et al. Long-term follow-up in children with benign convulsions associated with gastroenteritis. Eur J Paediatr Neurol 2014;18:572-7.


31. Chen B, Cheng M, Hong S, Liao S, Ma J, Li T, et al. Clinical outcome of recurrent afebrile seizures in children with benign convulsions associated with mild gastroenteritis. Seizure 2018;60:110-4.


32. Choi UY, Lee SY, Ma SH, Jang YT, Kim JY, Kim HM, et al. Epidemiological changes in rotavirus gastroenteritis in children under 5 years of age after the introduction of rotavirus vaccines in Korea. Eur J Pediatr 2013;172:947-52.



33. Choe YJ, Yang JJ, Park SK, Choi EH, Lee HJ. Comparative estimation of coverage between national immunization program vaccines and non-NIP vaccines in Korea. J Korean Med Sci 2013;28:1283-8.



34. Fischer TK, Ashley D, Kerin T, Reynolds-Hedmann E, Gentsch J, Widdowson MA, et al. Rotavirus antigenemia in patients with acute gastroenteritis. J Infect Dis 2005;192:913-9.



35. Blutt SE, Kirkwood CD, Parreno V, Warfield KL, Ciarlet M, Estes MK, et al. Rotavirus antigenaemia and viraemia: a common event? Lancet 2003;362:1445-9.


36. Weclewicz K, Svensson L, Kristensson K. Targeting of endoplasmic reticulum-associated proteins to axons and dendrites in rotavirus-infected neurons. Brain Res Bull 1998;46:353-60.



37. Yeom JS, Kim YS, Jun JS, Do HJ, Park JS, Seo JH, et al. NSP4 antibody levels in rotavirus gastroenteritis patients with seizures. Eur J Paediatr Neurol 2017;21:367-73.


38. Kawamura Y, Ohashi M, Ihira M, Hashimoto S, Taniguchi K, Yoshikawa T. Nationwide survey of rotavirus-associated encephalopathy and sudden unexpected death in Japan. Brain Dev 2014;36:601-7.


- TOOLS
-
METRICS

- Related articles in Ann Child Neurol
-
Clinical Investigation of Benign Afebrile Seizure with Gastroenteritis.2005 May;13(1)
A Clinical Study of Benign Convulsions Associated with Acute Gastroenteritis.2006 May;14(1)